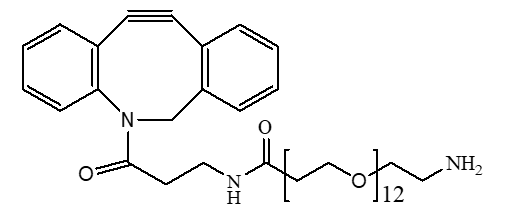
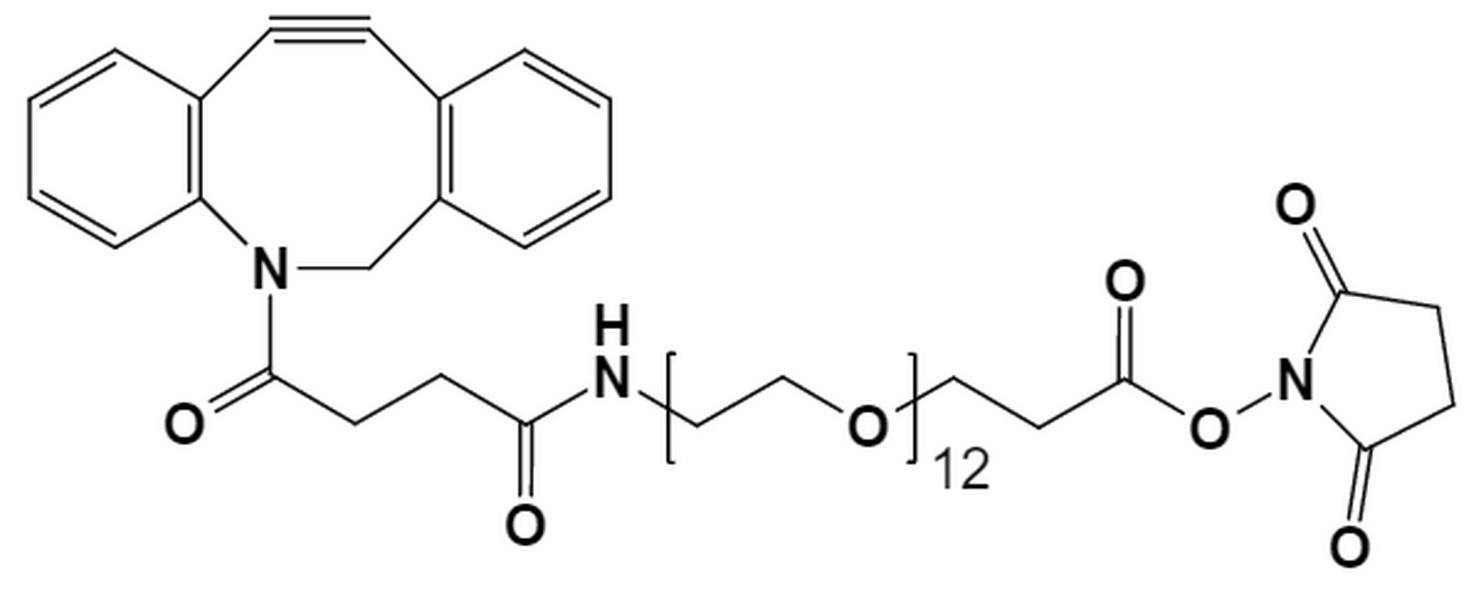
Antibody-Drug Conjugates (ADCs) represent a significant leap forward in targeted cancer therapy, combining the specificity of monoclonal antibodies with the potent cell-killing power of cytotoxic drugs. Polyethylene glycol (PEG) has emerged as a gold-standard material for linkers in ADC development. Its hydrophilic nature helps overcome the common issue of drug aggregation, enhancing the solubility and stability of the entire conjugate.
This paper will systematically introduce what an ADC is, the role of PEG in ADCs, the application advantages of PEG in ADCs, and future challenges.
What Are Antibody-Drug Conjugates (ADCs)?
Antibody-Drug Conjugates (ADCs) are a class of targeted biopharmaceutical agents designed for cancer treatment. By utilizing monoclonal antibodies that specifically recognize tumor-associated antigens, ADCs deliver highly potent cytotoxic drugs directly to cancer cells. This targeted approach enhances therapeutic efficacy while significantly reducing the systemic toxicity associated with conventional chemotherapy.
An ADC consists of three core components: a monoclonal antibody that binds to specific antigens on the surface of cancer cells, a highly toxic payload (which is too potent to be administered alone), and a linker that connects the two. The linker must remain stable during circulation in the bloodstream but efficiently release the drug upon reaching the target cells. As a critical element in ADC development, a well-designed linker with controlled release properties is essential to ensuring both targeting precision and treatment safety.
At PurePEG, we specialize in creating these critical components, offering a wide range of PEGylation Reagents and linkers to support ADC research.
The Role of PEG Linkers in Antibody-Drug Conjugates
PEG linkers are more than just a simple connection. They are sophisticated tools that modulate the physicochemical and pharmacokinetic properties of the ADC, directly contributing to its success as a therapeutic agent.
PEG linkers are versatile structures that can be designed with different functional groups at each end. These groups allow for specific conjugation chemistry, creating a stable bond between the antibody and the drug. For example, a Heterobifunctional PEG can have one end that reacts with an amino acid on the antibody (like lysine or cysteine) and another end that attaches to the drug molecule. This dual functionality is essential for controlled and efficient ADC construction.
PurePEG offers a range of linkers ideal for this purpose, including Cleavable Linkers that can be engineered for specific release mechanisms.
Benefits of PEG Linkers in ADC Development
Improving Solubility,Stability, and Targeting
Many potent cytotoxic drugs are hydrophobic, meaning they don’t dissolve well in water or blood. Attaching them to an antibody can cause the resulting ADC to aggregate, rendering it ineffective. PEG linkers are highly hydrophilic, counteracting the drug’s hydrophobicity and improving the overall solubility of the conjugate. This prevents aggregation and ensures the ADC remains stable and active in circulation. The PEG chain also creates a protective hydrophilic cloud around the payload, shielding it from metabolic enzymes and reducing the chances of an unwanted immune response.
Enhanced circulation time of therapeutic agents
The PEG chain increases the hydrodynamic size of the ADC, which slows its clearance by the kidneys. This extended half-life means the ADC remains in the bloodstream longer, providing a greater opportunity to locate and bind to tumor cells. This is a crucial factor for treating solid tumors, where penetration and accumulation can be slow.
Reduced toxicity and improved safety profile
By ensuring the cytotoxic payload remains attached to the antibody until it reaches the tumor, PEG linkers significantly reduce off-target toxicity. The hydrophilic PEG shield also masks the drug from the immune system, lowering the risk of immunogenicity. This improved safety profile allows for higher, more effective doses to be administered to the patient.
Better pharmacokinetics and biodistribution
PEGylation leads to more predictable and favorable pharmacokinetic (PK) properties. The improved solubility and stability prevent premature metabolism and clearance, ensuring that more of the drug reaches its intended target. This results in better biodistribution, with higher concentrations of the ADC accumulating in tumor tissue and lower concentrations in healthy organs.
Our advanced linkers, such as those in the PROTAC category, are built on these principles to optimize drug delivery.
Featured Products
Challenges of Using PEG Linkers in ADCs
Stability and premature release issues
The core challenge in linker design is achieving the perfect balance of stability. The linker must be robust enough to survive in the bloodstream but labile enough to release the payload inside the cancer cell. An imperfect design can lead to premature drug release, causing systemic toxicity. This is where the “cleavable PEG linker synthesis” process becomes critical.
Manufacturing complexity
The synthesis and conjugation of PEG linkers to antibodies and drugs is a complex, multi-step process. Ensuring batch-to-batch consistency and purity is a significant production assurance.
At PurePEG, our high-purity, monodisperse PEGs, such as PEG45 and Homobifunctional PEGs, which provide the consistency and quality required for reproducible results in ADC manufacturing.
Regulatory considerations
As a key component of the final drug product, PEG linkers are subject to intense regulatory scrutiny. Regulatory agencies like the FDA require extensive data on the stability, purity, and characterization of the linker and the final ADC.
Future of PEG Linkers in ADC Technology
Emerging drug research and innovations
Researchers are exploring novel linker designs that offer more precise control over drug release. This includes linkers that respond to multiple triggers or those that release the payload at a controlled rate. Furthermore, site-specific conjugation techniques are becoming more common. Products like our Clickable Linkers .
Trends shaping the future of targeted cancer therapies
The future of ADCs lies in greater precision. This includes developing ADCs for new targets, using novel payloads with different mechanisms of action, and engineering linkers that are tailored to the specific biology of a tumor. The overarching trend is moving toward personalized medicine, where the ADC design, including the “PEG linker for ADCs,” is optimized for an individual patient’s cancer type.
Conclusion: Why PEG Linkers Are Crucial for ADCs
PEG linkers are a foundational technology in the world of Antibody-Drug Conjugates. Their role goes far beyond simply connecting a drug to an antibody; they are enabling components that dictate the success or failure of a targeted therapy.By improving solubility, increasing stability, extending circulation time, and reducing toxicity, PEG linkers transform potent drugs and targeted antibodies into a single, highly effective therapeutic.
As oncology moves further into the era of precision medicine, the demand for sophisticated and reliable components will only grow. High-quality, well-characterized PEG linkers are essential tools for developing the next generation of ADCs that promise to be even more targeted and effective.