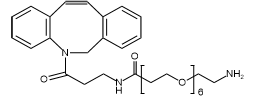
PurePEG is committed to providing the highest quality PEG linkers, backed by the documentation you need to confidently advance your project from preclinical research to commercial manufacturing.
The Importance of Regulatory Compliance in PEG Linker Sourcing
Regulatory agencies like the FDA and EMA demand rigorous characterization and control over every raw material used in a therapeutic product. For PEG linkers, this means demonstrating:
- Purity & Identity: Verifying the precise chemical structure and absence of impurities.
- Consistency: Ensuring that every batch produced meets the exact same specifications.
- Traceability: Documenting the entire manufacturing history, from raw materials to final product.
- Safety: Providing data to support the safe use of the material in a clinical setting.
Failure to provide adequate documentation can lead to significant delays, costly rework, and potential rejection of a regulatory filing. Partnering with a supplier who provides a thorough support package is essential for mitigating these risks.

What’s Inside PurePEG’s Regulatory Support Package?
1. Certificate of Analysis (CoA)
This document certifies that a specific batch of product meets all established quality specifications.
- Identity Verification: Confirmed by methods such as NMR and Mass Spectrometry.
- Purity Assessment: Detailed results from HPLC analysis, confirming purity levels often exceeding 99%.
- Key Specifications: Lot number, manufacturing date, and retest date.
2. Safety Data Sheet (SDS)
Provides comprehensive information on the substance for safe handling, storage, and disposal. It includes but is not limited to:
- CHEMICAL PRODUCT AND COMPANY IDENTIFICATION
- FIRST AID MEASURES
- HANDLING AND STORAGE
- PHYSICAL AND CHEMICAL PROPERTIES
- STABILITY AND REACTIVITY
- DISPOSAL CONSIDERATIONS
- TRANSPORT INFORMATION
Featured Products
3. Detailed Manufacturing Records
To ensure full transparency and support your quality audits, we provide detailed information about our manufacturing process.
- Batch Manufacturing Records Summary: An overview of the key steps and controls in the production process.
- Raw Material Sourcing: Information on the origin and quality control of starting materials.
4. BSE/TSE Statement
A declaration confirming that the product is free from Bovine Spongiform Encephalopathy (BSE) and Transmissible Spongiform Encephalopathy (TSE), a standard requirement for all biopharmaceutical components.
Accelerate Your Regulatory Submission with Confidence
Don’t let documentation challenges slow your progress. PurePEG provides the essential assurance for the efficient advancement of your project.