Copper-Free SPAAC Reagents for Bioorthogonal Conjugation, ADCs, and Advanced Drug Delivery
DBCO (dibenzocyclooctyne) reagents are a cornerstone of modern click chemistry, enabling fast, selective, and copper-free conjugation through strain-promoted azide–alkyne cycloaddition (SPAAC). Their unique combination of speed, stability, and biological compatibility makes DBCO reagents indispensable in bioconjugation, antibody–drug conjugates (ADCs), targeted drug delivery, diagnostics, and nanomedicine.
At PurePEG, DBCO reagents are engineered on high-purity, monodisperse PEG scaffolds, delivering consistent performance from discovery through translational and commercial workflows.
This page is the canonical product category hub for DBCO reagents, designed to help you:
- Understand how DBCO click chemistry works
- Identify when DBCO is the right click handle
- Select the appropriate PEG length and functionality
- Apply DBCO reagents in ADCs, proteins, and nanoparticles
- Navigate quality and regulatory considerations
What Are DBCO Reagents?
DBCO reagents contain a strained cyclooctyne ring that reacts rapidly and selectively with azide functional groups without the need for a copper catalyst. This reaction — SPAAC — is one of the most widely adopted bioorthogonal reactions in chemical biology.
A typical DBCO reagent consists of:
- A DBCO strained alkyne
- A PEG spacer of defined length (optional but common)
- A second functional group or attachment point
This modular structure allows DBCO reagents to function as:
- Click handles
- Conjugation reagents
- Linker components
- Targeting intermediates
For a broader overview of click chemistry, see Click Chemistry Reagents
Why DBCO Is the Gold Standard for Copper-Free Click Chemistry
DBCO reagents are preferred in biological systems because they offer:
- Copper-free reaction conditions (no cytotoxicity)
- Fast reaction kinetics
- Excellent selectivity
- Compatibility with live cells and in vivo systems
These properties make DBCO ideal for applications where biological integrity must be preserved, such as therapeutic conjugates and in vivo labeling.
DBCO vs Other Click Chemistry Handles
Compared with alternatives:
- CuAAC requires copper and is unsuitable for many biological systems
- BCN offers smaller size but different kinetics
- Tetrazine systems provide ultra-fast reactions but require complementary dienophiles
DBCO offers the best balance of speed, stability, and commercial maturity for most therapeutic and translational workflows.
Compare alternatives: BCN Reagents
Common Applications of NHS Ester PEG Reagents
Antibody–Drug Conjugates (ADCs)
DBCO reagents are increasingly used in ADC development to enable:
- Site-specific conjugation
- Controlled drug-to-antibody ratios (DAR)
- Reduced heterogeneity
- Improved stability
DBCO click chemistry allows ADC assembly without exposing antibodies to harsh conditions or metal catalysts.
Related resources:
Protein & Peptide Labeling
DBCO reagents enable:
- Rapid labeling of azide-modified proteins
- Site-specific attachment of probes
- High signal-to-noise labeling
This is particularly valuable in imaging, diagnostics, and mechanistic studies.
Nanomedicine & Targeted Drug Delivery
In nanoparticle and nanomedicine systems, DBCO reagents are used to:
- Attach targeting ligands
- Build modular delivery systems
- Assemble PEGylated nanocarriers
Explore related platforms:

Related Products
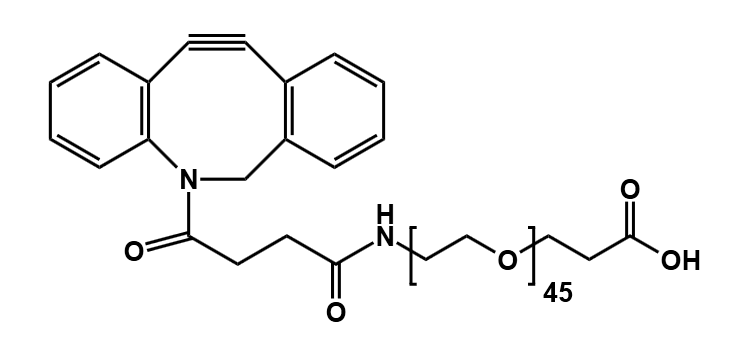
DBCO-CONH-PEG4-CH2CH2COOH
CAS# 1537170-85-6
MW# 552.61
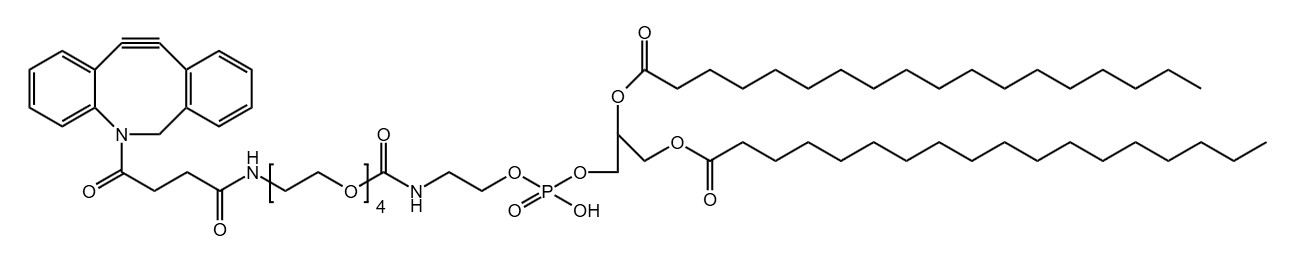
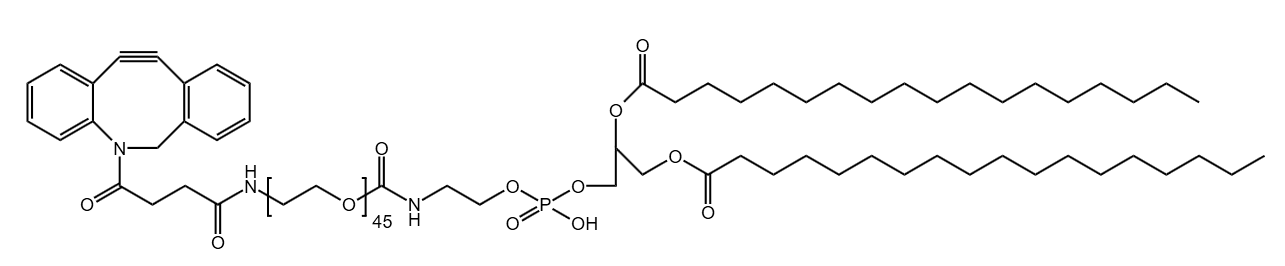
DBCO-CONH-PEG8-CH2CH2CO-DSPE
CAS# N/A
MW# 1458.87

Structural Variants of DBCO Reagents
PurePEG offers DBCO reagents across a wide range of functional formats.
DBCO-PEG Reagents
These incorporate DBCO onto a PEG backbone to:
- Improve solubility
- Reduce aggregation
- Provide controlled spacing
Common formats include:
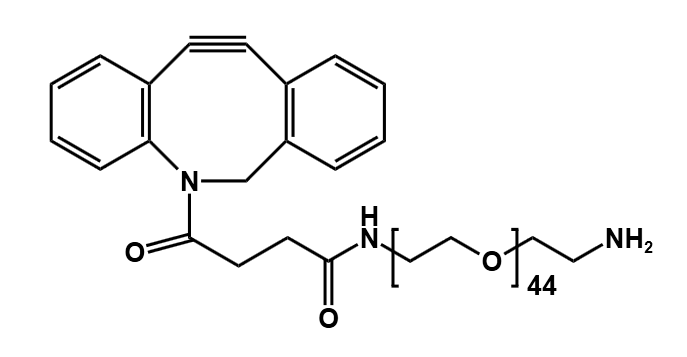
- DBCO-PEG-NH₂
- DBCO-PEG-COOH
- DBCO-PEG-NHS
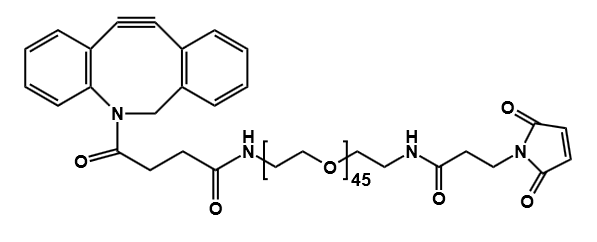
- DBCO-PEG-Maleimide
For PEG backbone fundamentals, see PEG Reagents & Building Blocks
Heterobifunctional DBCO Reagents
These reagents pair DBCO with a second functional group, enabling stepwise, orthogonal assembly.
Typical pairings include:
- DBCO–NHS
- DBCO–Maleimide
- DBCO–Amine
For linker-based architectures, see PEG Linkers
PEG Length Selection for DBCO Reagents
PEG length influences:
- Reaction accessibility
- Steric shielding
- Conjugate solubility
Short PEGs (PEG2–PEG6)
- Compact
- High reactivity
- Minimal shielding
Medium PEGs (PEG8–PEG24)
- Balanced performance
- Common in ADCs and targeting
Long PEGs (PEG36–PEG48+)
- Enhanced solubility
- Improved circulation
- Reduced immune recognition
Selection guidance: Why PEG Chain Length Matters


Stability & Handling Considerations
DBCO reagents are generally stable under standard storage conditions but should be protected from:
- Excessive heat
- Prolonged light exposure
Proper handling ensures optimal click reaction performance.
Quality & Regulatory Considerations
For therapeutic and translational use, DBCO reagents must meet strict standards for:
- Purity
- Structural definition
- Batch consistency
- Documentation
PurePEG supports:
- Monodisperse PEG-based DBCO reagents
- Controlled synthesis
- Regulatory-ready supply
Learn more: Regulatory Considerations


DBCO Reagents at PurePEG
This category includes:
- DBCO-PEG amines and acids
- DBCO-PEG NHS esters
- DBCO-PEG maleimides
- ADC-ready DBCO linkers
- Click-ready targeting intermediates
⬇️ Browse available DBCO products below
- dbco-peg*
- dbco-nh*
- dbco-biotin*
- dbco-val-cit-pab*

How to Choose the Right DBCO Reagent
When selecting a DBCO reagent, consider:
- Azide-bearing partner
- Desired conjugation speed
- PEG length requirements
- Secondary functional needs
- Downstream application and regulatory goals
For complex systems, PurePEG supports custom DBCO reagent synthesis and scale-up.
Explore Related Categories