Diazirine, Aryl Azide, and Photo-Crosslinking Reagents for Precision Bioconjugation
Photoreactive linkers enable covalent capture of transient biological interactions through light-activated crosslinking. They are essential tools in chemical biology, proteomics, diagnostics, drug discovery, and advanced bioconjugation, where conventional covalent chemistry cannot capture weak or dynamic interactions.
PurePEG’s photoreactive linkers combine high-purity photoreactive groups with monodisperse PEG spacers, delivering superior solubility, controlled reactivity, and reproducibility across research and translational workflows.
This page serves as the central product hub for photoreactive linker technologies, linking mechanism-level understanding, application guidance, and individual product SKUs.
What Are Photoreactive Linkers?
Photoreactive linkers are molecules that:
- Remain inert under normal conditions
- Become highly reactive upon UV or visible light activation
- Form covalent bonds with nearby biomolecules
They typically consist of:
- A photoreactive moiety
- A PEG spacer (optional but highly beneficial)
- One or more functional handles for conjugation
For PEG fundamentals, see PEG Reagents & Building Blocks
Why Use Photoreactive Linkers?
Photoreactive linkers enable researchers to:
- Capture weak or transient interactions
- Map protein–protein and protein–ligand contacts
- Study dynamic biological systems
- Improve conjugation specificity
They are indispensable in applications where:
- Binding is reversible
- Spatial proximity matters
- Traditional covalent chemistry fails
Major Classes of Photoreactive Linkers
Diazirine-Based Linkers
Diazirines are the most widely used photoreactive group due to:
- Small size (minimal steric disruption)
- Fast activation
- Broad reactivity with C–H, N–H, and O–H bonds
Upon UV irradiation, diazirines generate highly reactive carbenes that insert into nearby bonds.
Related category: Diazirine Linkers
Aryl Azide Linkers
Aryl azides generate nitrenes upon UV activation.
They offer:
- Strong reactivity
- Broad substrate compatibility
However, they are:
- Larger than diazirines
- More prone to side reactions
Benzophenone-Based Linkers
Benzophenones:
- Activate under longer wavelengths
- Enable repeated activation cycles
- Are bulkier than diazirines
Used when:
- Extended irradiation is required
- Larger crosslinking radius is acceptable


Role of PEG in Photoreactive Linkers
Incorporating PEG spacers provides:
- Improved aqueous solubility
- Reduced nonspecific binding
- Controlled spacing from the target
PEG length can be tuned to:
- Optimize accessibility
- Minimize steric interference
- Improve crosslinking efficiency
Selection guide: Why PEG Chain Length Matters
Related Products
5-(4-isothiocyanatophenyl)-10,15,20-(triphenyl)porphyrin
CAS# 863708-54-7
MW# 671.82
5,15-Di(4-aminophenyl)-10,20-diphenyl porphine
CAS# 116206-75-8
MW# 644.78
5,10,15,20-Tetrakis(4-aminophenyl)porphyrin
CAS# 22112-84-1
MW# 674.8
5,10,15,20-tetrakis(4-hydroxyphenyl)porphyrin
CAS# 51094-17-8
MW# 678.75
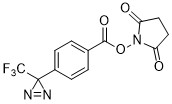
CF3-Diazirine-Ph-NHS ester
CAS# 87736-89-8
MW# 327.22
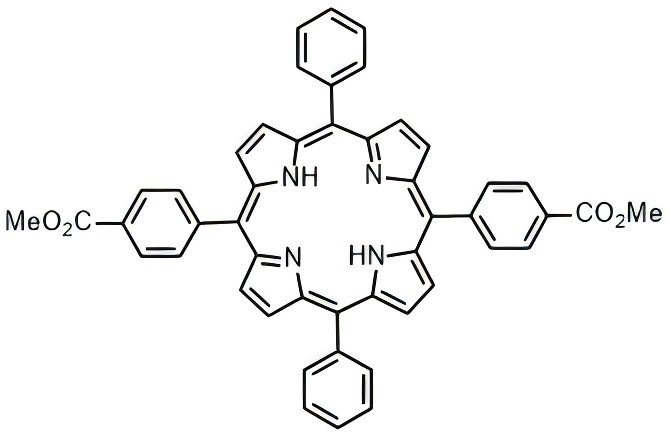
5,15-bis-(4-methoxycarbonylphenyl)-10,20-diphenylporphyrin
CAS# N/A
MW# 730.82
Applications of Photoreactive Linkers
Protein–Protein Interaction Mapping
Photoreactive linkers allow:
- Capture of transient complexes
- Identification of binding interfaces
- Structural biology insights
Used extensively in:
- Proteomics
- Signal transduction studies
Drug Discovery & Target Identification
Photoreactive probes enable:
- Identification of drug-binding partners
- Off-target profiling
- Mechanism-of-action studies
Related insight: Transforming ADC Viability
Antibody & Bioconjugation Applications
Photoreactive linkers can be used to:
- Attach probes without predefined reactive sites
- Capture antibody–antigen interactions
- Enable spatially controlled conjugation
Explore ADC integration: Antibody–Drug Conjugates
Diagnostics & Imaging
Photoreactive linkers are widely used in:
- Diagnostic assay development
- Imaging probe stabilization
- Biosensor construction
Related platform: Diagnostic Tools


Photoreactive Linkers vs Traditional Linkers
| Feature | Photoreactive Linkers | Traditional PEG Linkers |
| Activation | Light-triggered | Chemical |
| Specificity | Proximity-based | Functional group-based |
| Capture | Transient | Stable |
| Complexity | Higher | Lower |
Combining Photoreactive Linkers with Click Chemistry
Photoreactive linkers are often paired with:
- DBCO (SPAAC)
- BCN reagents
- Azide-functionalized targets
This enables:
- Modular probe assembly
- Orthogonal conjugation strategies
Explore compatible tools:


Stability, Storage, and Handling
Photoreactive linkers should be:
- Stored protected from light
- Handled under low-light conditions
- Activated only when ready for crosslinking
PurePEG provides:
- Stability data
- Handling guidance
- Technical documentation
Quality & Purity Considerations
High-quality photoreactive linkers require:
- Controlled photoreactive group integrity
- Monodisperse PEG backbones
- Minimal impurities
Learn more: High-Purity PEGs for Research


Photoreactive Linkers at PurePEG
This category includes:
- Diazirine-PEG linkers
- Aryl azide PEG linkers
- Photoreactive click-compatible linkers
- Custom photoreactive PEG designs
⬇️ Browse photoreactive linker products below
- diazirine*
- photo-crosslinkers
- porphyrins

How to Choose the Right Photoreactive Linker
Key selection criteria:
- Photoreactive group (diazirine vs azide vs benzophenone)
- PEG length
- Conjugation chemistry
- Application context
- Light source compatibility
PurePEG supports custom photoreactive linker synthesis for advanced workflows.
Explore Related Categories