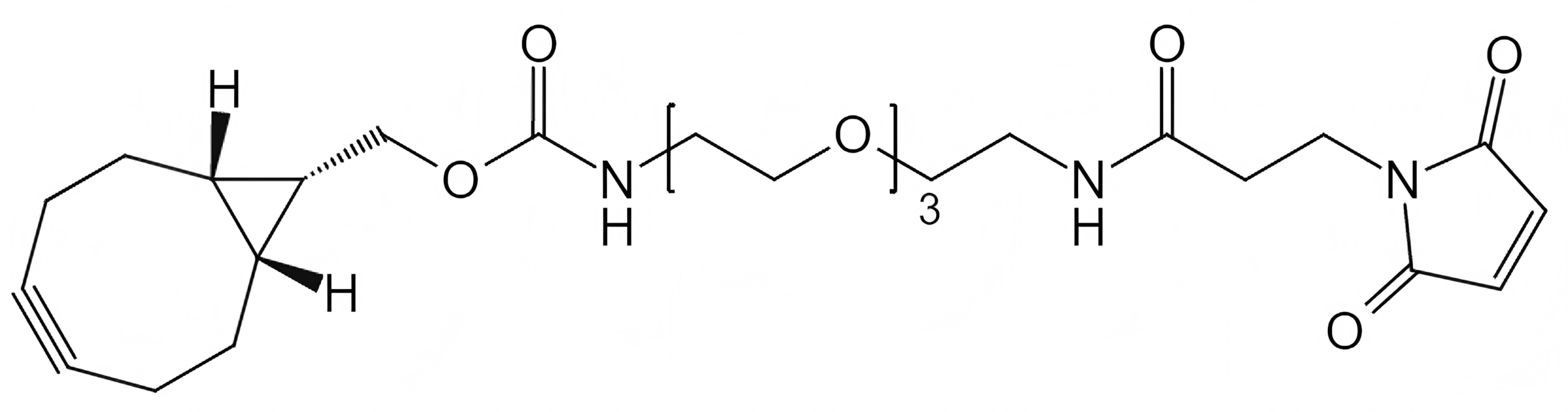
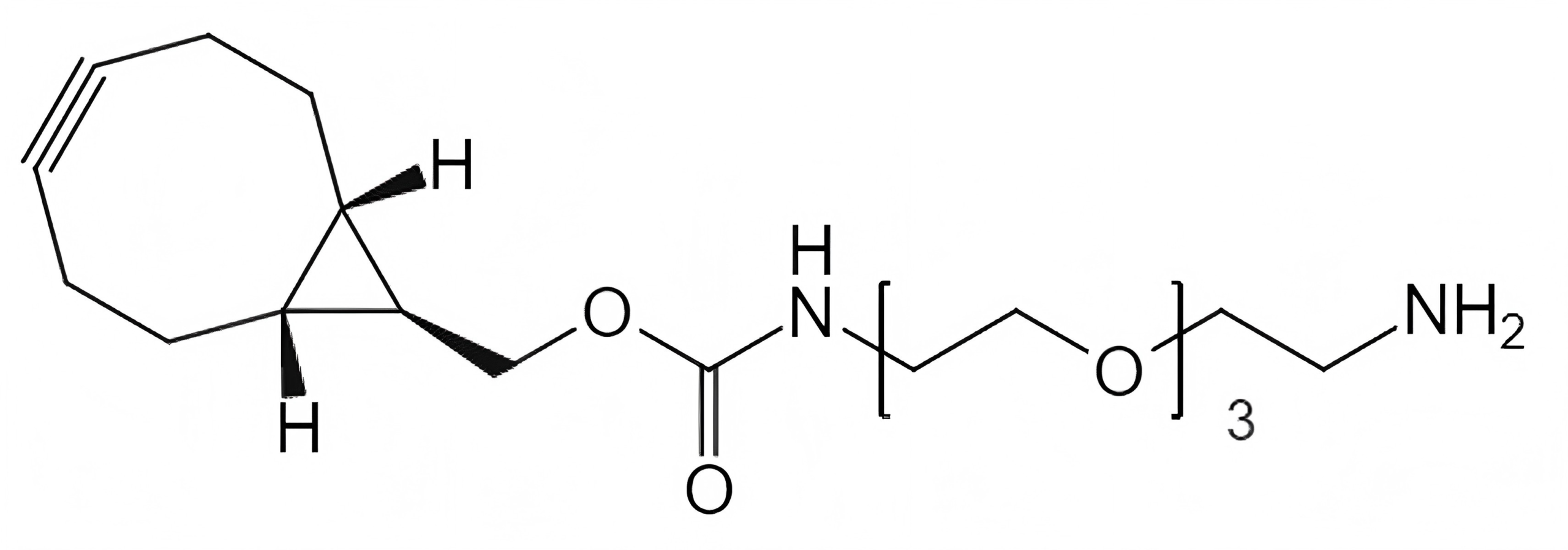
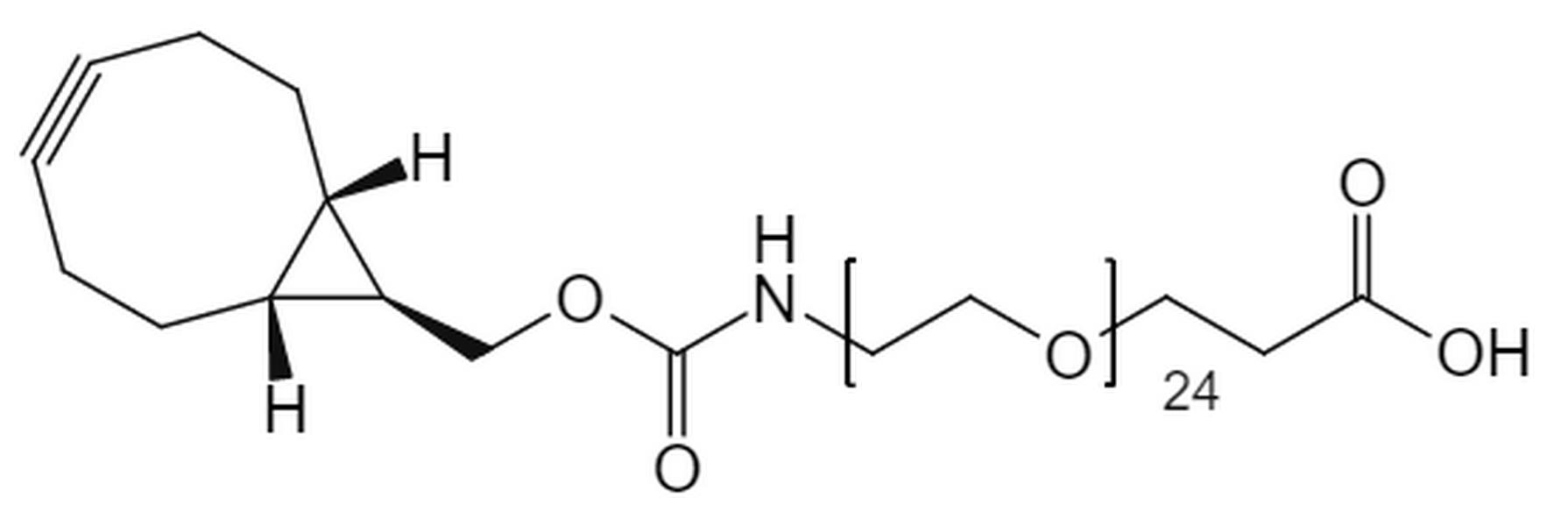
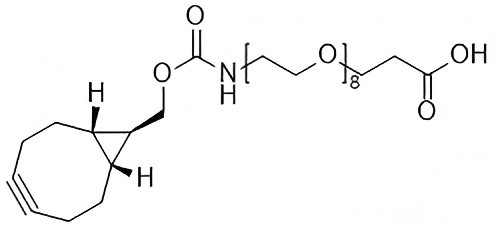
Strain-Promoted Click Chemistry Reagents for Fast, Copper-Free Bioconjugation
BCN (bicyclo[6.1.0]nonyne) reagents are high-performance, copper-free click chemistry tools used in bioorthogonal conjugation, ADC assembly, targeted drug delivery, diagnostics, and chemical biology. Their compact structure and rapid reaction kinetics make BCN reagents an attractive alternative or complement to DBCO-based systems.
PurePEG’s BCN reagents are engineered on high-purity, monodisperse PEG backbones, delivering predictable performance, excellent solubility, and scalability from discovery through translational development.
This page serves as the central product hub for BCN reagents, connecting mechanism-level understanding, application guidance, and individual SKU selection.
What Are BCN Reagents?
BCN reagents contain a strained cyclooctyne derivative that reacts selectively with azide-functionalized molecules via strain-promoted azide–alkyne cycloaddition (SPAAC) — without requiring copper catalysts.
Structurally, a BCN reagent typically includes:
- A BCN strained alkyne
- An optional PEG spacer
- A second functional handle for conjugation or attachment
BCN reagents are valued for their:
- Fast reaction rates
- Small steric footprint
- High bioorthogonality
For a general overview, see Click Chemistry Reagents
BCN vs DBCO: Key Differences
Both BCN and DBCO support copper-free click chemistry, but they differ in important ways.
| Feature | BCN Reagents | DBCO Reagents |
| Molecular size | Smaller | Larger |
| Steric bulk | Lower | Higher |
| Reaction speed | Fast | Very fast |
| Hydrophobicity | Lower | Higher |
| ADC compatibility | Excellent | Excellent |
BCN is often preferred when:
- Steric hindrance must be minimized
- Compact linker design is critical
- High labeling density is required
Compare directly: DBCO Reagents

Why Use BCN Reagents?
BCN reagents are widely used because they:
- Enable copper-free conjugation
- Preserve biomolecule integrity
- Perform well in live-cell and in vivo systems
- Integrate seamlessly with PEG-based architectures
They are especially valuable in protein, antibody, and nanoparticle conjugation workflows.
Role of PEG in BCN Reagents
PEGylation dramatically improves BCN reagent performance by:
- Increasing aqueous solubility
- Reducing aggregation
- Improving accessibility to azide targets
PurePEG offers BCN reagents with:
- Short PEG spacers (PEG2–PEG6)
- Medium PEG spacers (PEG8–PEG24)
- Long PEG spacers (PEG36–PEG45+)
PEG fundamentals: Why PEG Chain Length Matters

Related Products
endo-BCN-PEG3-Mal
CAS# 2141976-33-0
MW# 519.6
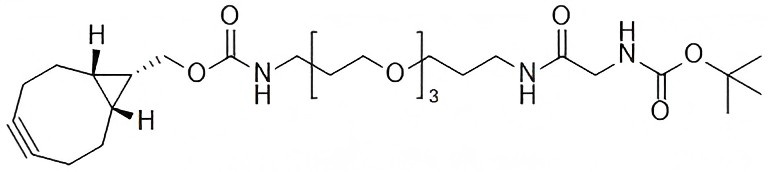
Boc-Gly-PEG3-endo-BCN
CAS# 2110444-63-6
MW# 553.7
Gly-PEG3-endo-BCN TFA salt
CAS# 2354291-37-3
MW# 453.6

Functional Formats of BCN Reagents
BCN-PEG-Amine & BCN-PEG-Acid
Used for:
- General bioconjugation
- Surface functionalization
- Stepwise linker assembly
BCN-PEG-Maleimide
Ideal for:
- Thiol-selective conjugation
- Cysteine-engineered antibodies
- Controlled ADC construction
Related chemistry: Maleimide PEG Linkers
BCN-PEG-NHS Esters
Used for:
- Amine coupling
- Protein and peptide labeling
- Rapid conjugate formation
Related category: NHS Ester PEG Reagents
BCN-PEG Click-Ready Linkers
BCN reagents are frequently combined with:
- Cleavable PEG motifs
- Photoreactive groups
- Payload-linker systems
Explore advanced architectures: Cleavable PEG Linkers
Photoreactive Linkers
Applications of BCN Reagents
Antibody–Drug Conjugates (ADCs)
BCN reagents enable:
- Site-specific conjugation
- Controlled DAR
- Reduced heterogeneity
Their compact size is especially beneficial in next-generation ADC designs.
ADC insights:
Protein & Peptide Labeling
BCN reagents are widely used for:
- Fluorophore attachment
- Affinity probe construction
- Imaging applications
They allow rapid, selective labeling under mild conditions.
Nanomedicine & Targeted Delivery
In nanoparticle systems, BCN reagents support:
- Ligand attachment
- Modular surface functionalization
- Controlled orientation of targeting moieties
Related platforms:
Diagnostics & Biosensors
BCN reagents are used to:
- Immobilize biomolecules
- Stabilize assay components
- Enable site-controlled labeling
Related area: Diagnostic Tools


BCN Reagents vs Traditional Conjugation Chemistry
| Feature | BCN Click Chemistry | Traditional Coupling |
| Catalyst required | No | Often |
| Selectivity | Very high | Moderate |
| Biocompatibility | Excellent | Variable |
| Speed | Fast | Variable |
Stability, Storage, and Handling
BCN reagents should be:
- Stored dry and protected from light
- Handled under standard laboratory conditions
- Used promptly after reconstitution
PurePEG provides stability data and handling guidance for all BCN products.


Quality & Regulatory Considerations
For translational and clinical workflows, BCN reagents must offer:
- Defined structure
- Controlled impurities
- Batch-to-batch consistency
PurePEG supports:
- Monodisperse PEG-based BCN reagents
- Scalable synthesis
- Regulatory documentation
Learn more: Regulatory Considerations
BCN Reagents at PurePEG
This category includes:
- BCN-PEG amines and acids
- BCN-PEG NHS esters
- BCN-PEG maleimides
- BCN-cleavable linker systems
- Custom BCN reagent designs
⬇️ Browse BCN reagent products below
- exo-bcn*
- endo-bcn*
- bcn-peg*


How to Choose the Right BCN Reagent
Key selection factors:
- Azide partner chemistry
- Required steric profile
- PEG length
- Secondary functional needs
- Application and regulatory stage
PurePEG offers custom BCN reagent synthesis and optimization.
Explore Related Categories