Thiol-Reactive Polyethylene Glycol Reagents for Site-Specific Bioconjugation, ADCs, and Drug Delivery
Maleimide PEG reagents are among the most widely used and trusted chemistries in modern bioconjugation. Their ability to react selectively with thiols enables highly controlled, site-specific conjugation, making them indispensable in applications ranging from antibody–drug conjugates (ADCs) to protein modification, nanomedicine, and targeted drug delivery.
At PurePEG, maleimide PEGs are engineered using high-purity, monodisperse PEG backbones, delivering reproducibility, scalability, and regulatory alignment across research, preclinical, and commercial programs.
This page serves as the central category hub for all maleimide PEG reagents, helping you understand:
- How maleimide PEG chemistry works
- When and why to use maleimide PEGs
- How PEG length and structure affect performance
- The role of maleimide PEGs in ADCs and advanced therapeutics
- How to select the right maleimide PEG for your application
What Are Maleimide PEG Reagents?
Maleimide PEG reagents are thiol-reactive PEG derivatives containing a maleimide functional group capable of forming stable thioether bonds with cysteine residues.
The general structure consists of:
- A maleimide reactive group
- A PEG spacer of defined length
- An optional second functional group (for heterobifunctional systems)
This architecture allows maleimide PEGs to act as:
- Conjugation reagents
- Molecular spacers
- Linker components
- Solubility enhancers
For an overview of PEG functional groups, see Functionalized PEGs
Why Maleimide PEG Chemistry Is So Widely Used
Maleimide–thiol chemistry is favored because it offers:
- High selectivity for thiols over amines
- Fast reaction kinetics under mild conditions
- Minimal side reactions
- Stable conjugates suitable for biological systems
This makes maleimide PEGs ideal for:
- Site-specific protein modification
- Controlled antibody conjugation
- High-value therapeutic constructs
Maleimide PEGs vs Other Thiol-Reactive Chemistries
Compared to alternatives such as haloacetyls or vinyl sulfones, maleimide PEGs offer a balance of:
- Speed
- Selectivity
- Stability
- Commercial precedent
For applications where controlled conjugation and reproducibility are critical, maleimide PEGs remain the gold standard.
Common Applications of Maleimide PEG Reagents
Antibody–Drug Conjugates (ADCs)
Maleimide PEGs are foundational in ADC development, where precise drug-to-antibody ratios (DAR) and conjugation homogeneity are critical.
Key roles include:
- Linking payloads to engineered cysteine residues
- Improving payload solubility
- Reducing aggregation
- Enhancing pharmacokinetics
Related resources:
Antibody–Drug Conjugates
PEG Linkers in ADCs
Protein & Peptide Conjugation
Maleimide PEGs enable:
- Site-specific labeling
- PEGylation of cysteine-containing proteins
- Enzyme stabilization
- Improved solubility and half-life
Nanomedicine & Drug Delivery
In nanoparticle and nanomedicine systems, maleimide PEGs are used to:
- Attach targeting ligands
- Control surface architecture
- Improve circulation time
- Reduce immune recognition
Explore related systems:
PEG Linkers
PEG Lipids


Maleimide PEG Structural Variants
PurePEG offers maleimide PEGs across a wide range of structural formats.
Monofunctional Maleimide PEGs
These feature:
- One maleimide group
- One inert terminus (e.g., methoxy)
Use cases:
- Single-point conjugation
- PEGylation
- Surface modification
Heterobifunctional Maleimide PEGs
These include maleimide paired with a second reactive group, such as:
- NHS ester
- Azide
- DBCO
- Amine
- Carboxyl
These reagents enable stepwise, orthogonal conjugation.
For advanced architectures, see PEG Linkers
Related Products
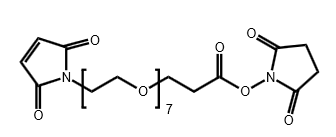
Maleimide-PEG7-CH2CH2COONHS Ester
CAS# N/A
MW# 574.57
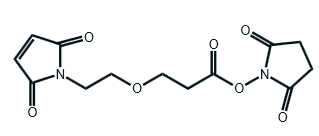
Maleimide-PEG1-CH2CH2COONHS Ester
CAS# 1807518-72-4
MW# 310.26
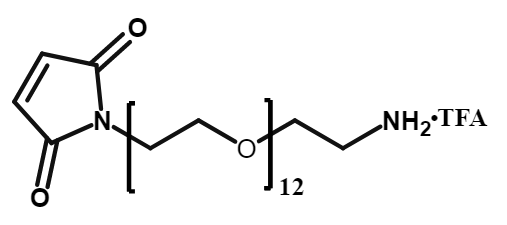
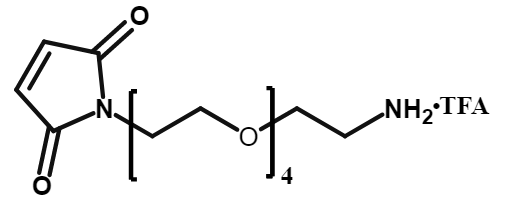
Mal-PEG12-CH2CH2NH2•TFA
CAS# 2170654-81-4
MW# 668.77
Mal-PEG4-CH2CH2NH2•TFA
CAS# 2221042-92-6
MW# 316.35
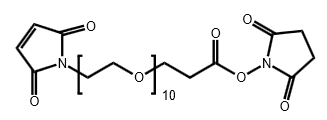
Maleimide-PEG10-CH2CH2COONHS Ester
CAS# N/A
MW# 706.73
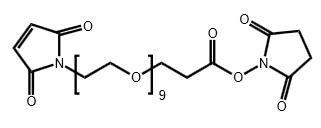
Maleimide-PEG9-CH2CH2COONHS Ester
CAS# N/A
MW# 662.68
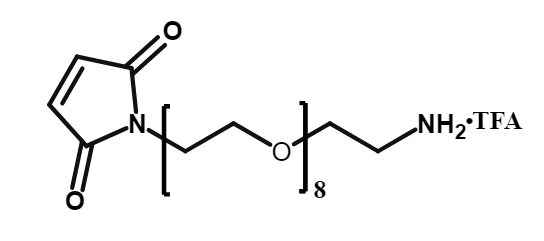
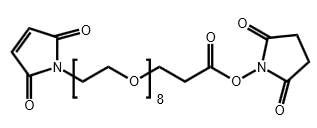
Maleimide-PEG8-CH2CH2COONHS Ester
CAS# 2055033-05-9
MW# 618.63
PEG Length Selection for Maleimide PEGs
PEG length significantly impacts conjugate behavior.
Short PEGs (PEG2–PEG6)
- Minimal spacing
- High reactivity
- Limited steric shielding
Medium PEGs (PEG8–PEG24)
- Balanced flexibility
- Common in ADCs
- Improved solubility
Long PEGs (PEG36–PEG48+)
- Enhanced stealth
- Improved circulation
- Reduced immune recognition
Selection guidance: Why PEG Chain Length Matter


Stability Considerations for Maleimide PEG Conjugates
While maleimide–thiol bonds are generally stable, factors such as:
- pH
- Thiol exchange
- In vivo environment
can influence long-term stability.
For applications requiring controlled release, cleavable PEG linker systems may be preferred.
See alternatives: Cleavable PEG Linkers
Quality & Regulatory Considerations
For therapeutic and translational use, maleimide PEGs must meet high standards for:
- Purity
- Functionalization efficiency
- Batch consistency
- Documentation
PurePEG emphasizes:
- Monodisperse PEG backbones
- Controlled synthesis
- Reproducible performance
- Regulatory-ready support
Learn more: Regulatory Considerations


Maleimide PEG Products at PurePEG
This category includes:
- Maleimide PEG acids
- Maleimide PEG amines
- Maleimide PEG NHS esters
- Maleimide PEG click-ready reagents
- ADC-compatible maleimide linkers
Browse available Maleimide PEG products below
- maleimide-peg3-*
- maleimide-peg6-*
- maleimide-peg10-*
- maleimide-nh-peg*
- mal-peg*-nh-mal
- maleimide-peg*-nhs-ester

How to Choose the Right Maleimide PEG
When selecting a maleimide PEG reagent, consider:
- Target molecule (protein, antibody, nanoparticle)
- Available thiol sites
- Desired PEG length
- Need for secondary functionality
- Downstream application and regulatory path
If you need help selecting or customizing a maleimide PEG, PurePEG’s scientific team can support custom synthesis and scale-up.