Antibody-Drug Conjugates (ADCs) represent a powerful class of targeted therapeutics, combining the specificity of monoclonal antibodies with the potent cytotoxicity of small-molecule drugs. The clinical success of an ADC is profoundly influenced by one of its most critical yet often overlooked components: the linker. This molecular bridge not only connects the antibody to the payload but also precisely controls the release mechanism of the active drug. The strategic decision between employing a cleavable or a non-cleavable polyethylene glycol (PEG) linker directly impacts the ADC’s stability, efficacy, safety, and therapeutic index.
This guide provides a comparative analysis of cleavable and non-cleavable PEG linkers in ADC design. We will examine their distinct mechanisms of action, key properties, and the decisive factors that inform the selection process, equipping your team to optimize the next generation of ADCs.

Introduction to PEG Linkers in ADCs
Overview of Antibody-Drug Conjugates (ADCs)
An ADC is complex biopharmaceutical composed of three essential elements:
- A monoclonal antibody that selectively binds to a tumor-associated antigen.
- An extremely potent cytotoxic payload.
- A chemical linker that covalently attaches the payload to the antibody.
The objective is to achieve targeted delivery of the cytotoxic agent to malignant cells, thereby maximizing antitumor activity while minimizing systemic exposure and off-target toxicity. The linker plays a pivotal role in maintaining the integrity of the conjugate during systemic circulation and ensuring precise payload release at the tumor site.
Importance of Linker Selection in ADC Performance
Linker selection is a critical determinant of ADC performance. It influences:
- Plasma Stability: Preventing premature payload release in the bloodstream.
- Pharmacokinetics: Modulating the ADC’s circulation half-life.
- Therapeutic Index: Balancing efficacy against target cells with safety in healthy tissues.
- Mechanism of Action: Dictating how and when the payload is activated.
The incorporation of a PEG spacer enhances solubility and reduces immunogenicity, but the core functionality—whether the linker is cleavable or non-cleavable—defines the ADC’s fundamental drug release strategy.
What Are Cleavable PEG Linkers?
Cleavable PEG linkers are engineered with strategically incorporated, labile bonds that remain stable in circulation but undergo specific cleavage upon exposure to unique conditions within the target cell (e.g., low pH, specific enzymes, or reducing environments). This design enables controlled intracellular drug release.
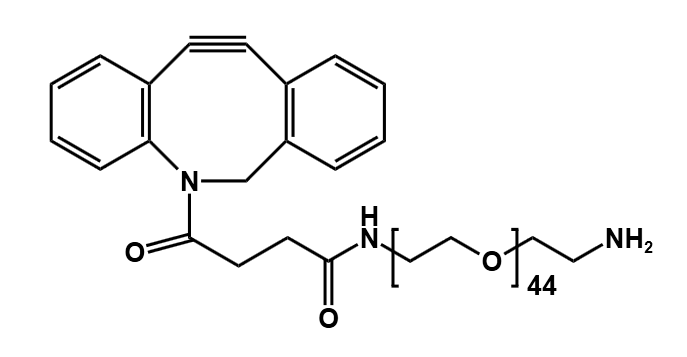
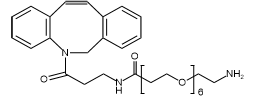
At PurePEG, our portfolio of Cleavable Linkers includes several types designed for specific triggers:
- Enzyme-Sensitive Linkers (e.g., Peptide-based): Contain sequences (such as Val-Ala, Val-Cit,) that are substrates for lysosomal proteases like cathepsin B, which is often overexpressed in tumors. Such as: MC-Val-Cit-PAB-PNP, Fmoc-Val-Ala-PAB-PNP.
- Reduction-Sensitive Linkers: such as Disulfide linkers, which are cleaved under high glutathione (GSH) conditions, and Diselenide linkers, which can be cleaved by both high concentrations of GSH and hydrogen peroxide (H₂O₂).
Mechanism of Payload Release in Target Cells
The process for cleavable linker-based ADCs follows a defined sequence:
1. Binding: The ADC binds to its target antigen on the cancer cell surface.
2. Internalization: The ADC-antigen complex is internalized via endocytosis.
3. Trafficking: The ADC is trafficked to acidic compartments (endosomes) and finally to lysosomes.
4. Cleavage and Release: The specific trigger (low pH, enzymes) within the lysosome cleaves the linker, releasing the active payload.
5. Cell Killing: The free payload exerts its cytotoxic effect by damaging essential cellular components.
Benefits for Controlled Drug Release
The primary advantage of cleavable linkers is the release of the native, unmodified payload, which is often essential for optimal potency. A significant additional benefit is the “bystander effect”: the released drug can diffuse across cell membranes and kill adjacent cancer cells that may not express the target antigen, enhancing efficacy against heterogeneous tumors.
Featured Products
What Are Non-Cleavable PEG Linkers?
Definition and chemical characteristics
Non-cleavable PEG linkers form a highly stable, covalent bond between the antibody and the payload that are designed to withstand extracellular and intracellular environments. Drug release occurs only after the antibody moiety is completely degraded within the lysosome, resulting in the release of the payload attached to a linker remnant (typically an amino acid residue).
Our Heterobifunctional PEGs and Homobifunctional PEGs can be used to create robust, non-cleavable constructs for applications where stability is paramount.
Stability Advantages and Pharmacokinetic Benefits
The principal advantage of non-cleavable linkers is their exceptional stability, which virtually eliminates the risk of premature payload release in plasma. This leads to:
- A significantly improved safety profile due to reduced systemic toxicity.
- A more predictable pharmacokinetic (PK) profile and extended circulation half-life.
- Simplified metabolite characterization.
Ideal Use Cases
Non-cleavable linkers are the preferred choice when:
- The payload-linker-amino acid metabolite retains high cytotoxic potency.
- The primary developmentgoal is to minimize off-target toxicity.
- A bystander effect is undesirable (e.g., to protect sensitive neighboring healthy tissues).
- The target antigen is is internalized slowly, necessitating a long circulatory half-life for optimal tumor accumulation.
Key Differences: Cleavable and Non-Cleavable PEG Linkers
Mechanism of Drug Release
- Cleavable Linkers: Release payload via a specific trigger-mediated cleavageinside the target cell.
- Non-Cleavable Linkers: Release payload as a cataboliteafter full proteolytic degradation of the antibody scaffold.
Impact on Pharmacokinetics and Therapeutic Index
- Cleavable ADCs: May exhibit more complex PK/PD due to the bystander effect and potential for minor premature release. Can achieve a high therapeutic index if tumor-specific release is efficient.
- Non-Cleavable ADCs: Typically demonstrate simpler, more predictable PK with a wider therapeutic window due to superior plasma stability.
Comparative Summary of Key Attributes
| Feature | Cleavable Linkers | Non-Cleavable Linkers |
| Plasma Stability | Stable in circulation but designed for controlled breakdown in target cells. Risk of premature cleavage exists. | Excellent. Highly stable in circulation with minimal risk of off-target release. |
| Safety Profile | Higher potential for off-target toxicity if premature cleavage occurs. Bystander effect can be dual-edged. | Generally superior safer due to minimized systemic exposure to free payload. |
| Efficacy Potential | High potency potential from release of native payload and bystander effect. | Efficacy contingent on the activity of the payload-amino acid metabolite.. |
| Tumor Heterogeneity | Can be advantageous (bystander effect). | Less effective against antigen-negative cells. |
Challenges and Considerations
Manufacturing Complexity
Synthesizing high-purity cleavable linkers requires precise control to ensure the labile bond is stable during manufacturing yet responsive in the biological environment. For all linkers, achieving monodispersity and rigorous batch-to-batch consistency is critical for clinical translation and regulatory approval.
Regulatory and Characterization Hurdles
Regulatory agencies require comprehensive data on ADC stability, metabolite profiles, and potential off-target effects. For cleavable linkers, extensive plasma stability studies are essential. For non-cleavable linkers, the potency and toxicology of the final metabolite must be thoroughly characterized.
Conclusion: Optimizing ADC Efficacy with Strategic Linker Selection
The choice between a cleavable and a non-cleavable PEG linker is a fundamental strategic decision in ADC design. Cleavable linkers offer the potential for enhanced potency through the bystander effect and release of the native drug. Non-cleavable linkers provide a superior safety profile driven by exceptional plasma stability. There is no universal best choice; the decision must be aligned with the specific attributes of the antibody, payload, target biology, and desired therapeutic outcome.
The linker is the crucial element that dictates the success of an ADC. A scientifically informed linker selection, supported by high-quality materials from a trusted supplier like PurePEG, is indispensable for developing safer and more effective targeted therapies.