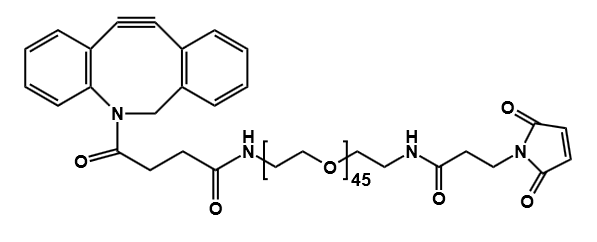
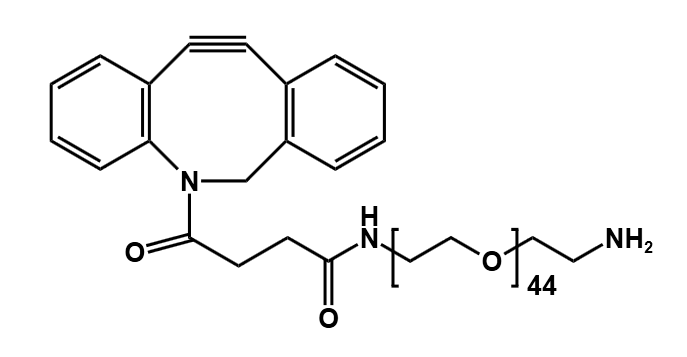
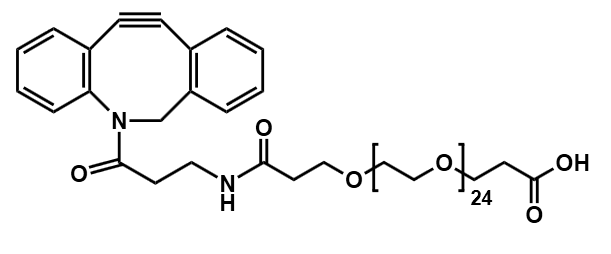
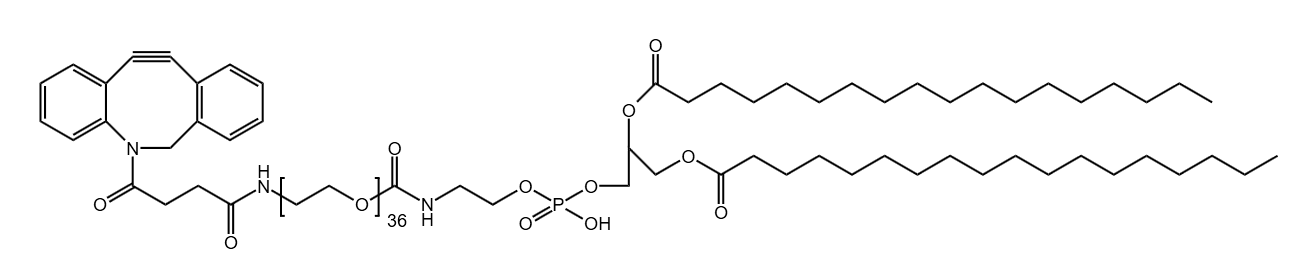
Antibody-drug conjugates (ADCs) have transformed the paradigm of targeted cancer therapy. By combining the specificity of monoclonal antibodies with the potent cytotoxicity of small-molecule drugs, ADCs enable the directed delivery of therapeutic agents to malignant cells, thereby minimizing damage to healthy tissues. The efficacy of this sophisticated modality is critically dependent on one component: the linker. This molecular bridge serves as the central control unit, precisely governing the stability, release kinetics, and overall therapeutic profile of the conjugate.
This overview examines the fundamentals of ADC linker technology, with a special focus on the essential role of polyethylene glycol (PEG) in creating safer and more effective therapies. We will cover the evolution of linkers from simple connectors to intelligent molecular tools, detail the different types available, and explain how they function to optimize targeted drug delivery.
The Evolution of ADC Linker Technology
The journey of ADC development has been one of continuous refinement, and linker technology has been at the center of this progress. Each generation has built upon the lessons of the last, moving toward the ultimate goal of maximizing efficacy while minimizing toxicity.
First-Generation Linkers: Facing Stability Challenges
Early linkers (such as non-cleavable types) exhibited insufficient stability in the systemic circulation, leading to premature release of cytotoxic payloads before reaching targets. This triggered severe off-target toxicity issues, significantly limiting their therapeutic window. This bottleneck underscored the necessity for systematic engineering of linkers.
Second-Generation Linkers: Enabling Targeted Controlled Release
To address stability issues, second-generation cleavable linkers emerged. They maintain high stability in the circulatory system while being selectively activated within specific intracellular environments—such as the low pH of lysosomes or specific proteases—enabling targeted controlled release of the payload. This breakthrough design—including acid-cleavable linkers and enzyme-cleavable peptide linkers—significantly reduced off-target toxicity, marking a critical step toward ADC maturity.
Third-Generation & Next-Generation Linkers: Advancing Precision and Multifunctionality
Current technological frontiers focus on precision and functional optimization. Next-generation linkers not only pursue higher tumor microenvironment specificity but also systematically improve ADC solubility, pharmacokinetic properties, and reduce immunogenicity through strategies like polyethylene glycol (PEG) modification. Furthermore, technologies aimed at producing uniformly distributed conjugates (uniform DAR) and smart linkers capable of responding to dual signals or precisely regulating the bystander effect are driving ADCs toward safer and more effective applications.
Linkers have evolved from simple chemical bridges into core functional modules within ADC design, enabling the achievement of optimal therapeutic indices.
Featured Products
The Critical Role of PEG Linkers in Modern ADCs
Polyethylene glycol (PEG) has become an indispensable tool in modern ADC design. These flexible, water-soluble chains are not just spacers; they offer a multitude of benefits that address many of the core challenges in developing effective and safe ADCs.
Enhancing Solubility and Conjugate Stability
Many of the most potent anticancer drugs are highly hydrophobic, meaning they do not dissolve well in water. Attaching these drugs to an antibody can cause the entire ADC to clump together, or aggregate, rendering it ineffective. Incorporating a PEG chain into the linker dramatically increases the ADC’s overall water solubility. This prevents aggregation and ensures the ADC remains stable and active in the bloodstream. Our monodisperse, high-purity PEGylation Reagents significantly enhance drug solubility, stability, and experimental reproducibility in vivo.
Optimizing Pharmacokinetic Profiles
The “pharmacokinetics” (PK) of a drug describe its absorption, distribution, metabolism, and excretion in the body. A key goal for ADC is to have a long circulation half-life, which gives it more time to locate and bind to its target cancer cells. The PEG chain creates a hydrophilic “cloud” around the ADC, which shields it from detection by the immune system and reduces clearance by the kidneys. This extended circulation time is a hallmark of well-designed, PEGylated ADCs.
Enabling Site-Specific Conjugation
Modern ADC development aims for a uniform drug-to-antibody ratio (DAR), meaning each antibody carries the same number of drug molecules. Attaching linkers at specific, engineered sites on the antibody leads to a homogeneous product with predictable behavior and a better safety profile. PEG linkers with specific reactive groups, such as our versatile Heterobifunctional PEGs and Clickable Linkers are essential for achieving this controlled, site-specific conjugation.
Linker Classification: Cleavable vs. Non-Cleavable
The most fundamental choice in linker design is whether it should break apart to release the drug (cleavable) or remain permanently attached (non-cleavable). This decision is critical and depends entirely on the payload’s mechanism of action.
Cleavable Linkers: Enabling Controlled Intracellular Activation
Cleavable linkers are designed to break and release the drug upon encountering a specific trigger inside or around the cancer cell. This is the most common strategy, as many payloads are only active in their original, unmodified form.
Common types of cleavable linkers include:
- Enzyme-Cleavable Linkers: These contain a short peptide sequence that is recognized and cut by enzymes, like cathepsin B, that are highly active inside cancer cell lysosomes. Linkers such as MC-Val-Cit-PAB-PNP are classic examples of this technology.
- Acid-Labile (pH-Sensitive) Linkers: These linkers (such as a hydrazone) remain stable under the neutral pH (~7.4) conditions of blood. However, once the ADC is internalized and enters acidic endosomes/lysosomes, the linkers undergo acid hydrolysis under low pH conditions (pH 5.0-6.5), leading to specific cleavage and release of the cytotoxic payload.
- Reduction-Sensitive Linkers:Such linkers remain stable in the oxidising environment of blood but are rapidly cleaved in the reducing environment of target cells, which harbour high concentrations of glutathione and hydrogen peroxide, thereby releasing the toxic or active payload. PurePEG offers a range of functional group linkers including disulfide linkers, disulfide PEG linkers, and diselenide linkers.
You can explore our full range of Cleavable Linkers engineered for precisely controlled drug release.
Non-Cleavable Linkers: Maximizing Plasma Stability
Non-cleavable linkers form a highly stable chemical bond that does not break. The drug is released only after the entire antibody is degraded by the cell’s internal machinery. In this case, the payload is released with the linker and a residual amino acid still attached. This approach is effective for payloads that remain active even with this small modification.
The primary advantage of non-cleavable linkers is their exceptional stability, which can lead to an improved safety profile by minimizing any chance of premature drug release. The best PEG linker for ADCs using this strategy is often a simple but robust heterobifunctional linker that provides necessary spacing and solubility without a cleavable element.
Applications in Advanced Targeted Therapeutics
Advanced linker technology is the enabler of precision-targeted delivery. By strategically selecting the linker, researchers can tailor ADC properties to the specific biology of a cancer type. For example, an ADC for a solid tumor might employ an enzyme-cleavable linker responsive to the tumor microenvironment, while an ADC for a hematologic malignancy might utilize a highly stable non-cleavable linker for sustained activity.
The versatility of PEG linkers allows for their use in many cutting-edge applications, including:
- Dual-Payload ADCs: Attaching two different types of drugs to the same antibody to attack cancer cells through multiple mechanisms. Multi-Arm PEGs are an enabling technology for these complex, multi-pronged designs.
- PROTAC Conjugates: Using antibodies to deliver protein degraders (PROTACs) to specific tissues for targeted protein removal. Our specialized PROTAC are designed for this cutting-edge application.
- Bioorthogonal Conjugation: Employing highly efficient and specific chemistries, such as copper-free click reactions, for gentle and controlled assembly. Our extensive library of Clickable Linkers provides robust solutions for modern bioconjugation..
Conclusion: The Strategic Imperative of Linker Design
The linker is the central functional element that dictates the ultimate success of an ADC, governing its safety, efficacy, and developability. As the field advances toward more complex and potent therapeutics, the demand for well-defined, high-purity, and functionally sophisticated linkers will continue to grow. PEG-based linkers have firmly established themselves as a cornerstone of modern ADC design, providing the critical properties needed to translate promising molecular concepts into clinically successful therapies.
A deep understanding of linker technology empowers researchers to make strategic decisions in designing the next generation of targeted oncology treatments.
Ready to build a better ADC? Contact our experts for custom synthesis support.