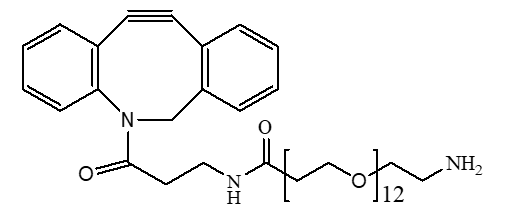
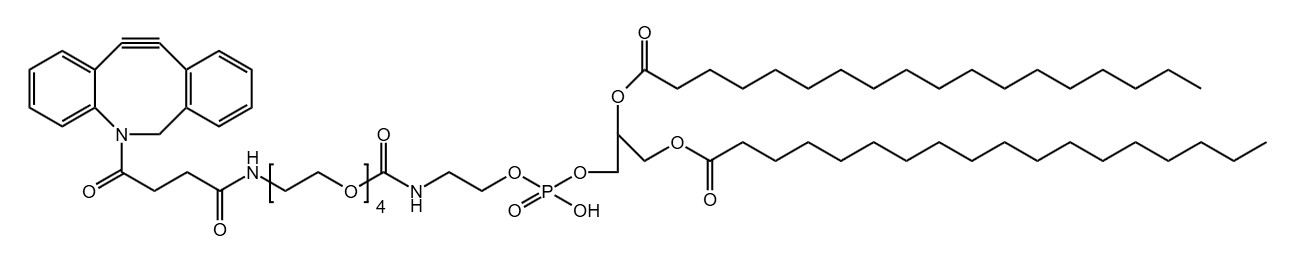
What Are Controlled-Release Drugs?
Controlled-release drugs represent a core direction in modern drug design, aiming to precisely regulate drug release behavior within the body through sophisticated engineering techniques. Compared to traditional formulations, their design objective is to enable sustained drug release at a predetermined rate over a specific timeframe. This maintains stable blood concentrations within the therapeutic window, effectively preventing efficacy inconsistencies or adverse reactions caused by concentration fluctuations.
This strategy significantly overcomes multiple barriers to drug delivery within the body, including enzymatic degradation, rapid clearance, and non-specific damage to healthy tissues. By sustaining steady effective concentrations, controlled-release drugs improve patient compliance, enhance therapeutic efficacy and safety, and provide more advantageous pharmaceutical solutions for treating complex conditions such as chronic diseases and tumors.
What Are PEG Linkers?
Polyethylene glycol (PEG) is a water-soluble polymer composed of repeating ethylene oxide units, widely utilized in biomedical fields due to its excellent biocompatibility, low immunogenicity, and non-toxicity. The core value of PEG linkers lies in their multifunctional bridging capability, enabling precise coupling of drug molecules with targeting ligands, carrier systems, or other functional components to construct efficient delivery systems. Both the molecular chain length and terminal functional groups can be flexibly designed and synthesized according to application requirements.
Types of PEG linkers used in pharmaceuticals
To serve different purposes, PEG linkers come in various forms.
- Heterobifunctional PEGs: These linkers have different reactive groups at each end, allowing for the sequential conjugation of two different molecules. They are often the best PEG linker for protein conjugation and creating complex bioconjugates like antibody-drug conjugates (ADCs).
- Homobifunctional PEGs: With identical reactive groups at both ends, these are used for crosslinking molecules or creating polymers.
- Cleavable Linkers: These are designed to break apart under specific physiological conditions, such as changes in pH or the presence of certain enzymes. This feature is crucial for releasing a drug precisely at the target site, like a tumor environment. The process of cleavable PEG linker synthesis allows for tailored release mechanisms.
- Clickable Linkers: These linkers utilize “click chemistry,” a set of highly efficient and specific reactions. Products like our DBCO-CONH-PEG45-CH2CH2COOH are perfect for bioorthogonal labeling and conjugation without interfering with biological functions.
Featured Products
How PEG Linkers Enable Controlled Release
Polyethylene glycol (PEG) linkers provide multiple critical functions for drug delivery systems through their unique physicochemical properties. Regarding controlled release mechanisms, PEG linkers can be engineered to achieve different release logics: cleavable linkers respond to specific biological signals (such as enzymes, pH, or reducing environments) within target sites like tumor cells, enabling site-specific drug release. This enhances therapeutic efficacy while significantly reducing systemic toxicity. Non-cleavable linkers, conversely, maintain drug-carrier structural integrity through stable covalent bonds. They rely on the hydration protective layer formed by PEG chains to delay renal clearance and shield against enzymatic degradation, thereby prolonging drug half-life and improving pharmacokinetic behavior.
Additionally, PEG linkers effectively address the water solubility challenges of drug molecules. Their highly hydrophilic nature forms a stable hydration layer on hydrophobic drug surfaces, significantly enhancing the solubility of the entire complex and formulation stability. This not only avoids the use of irritating organic solvents but also enables the development of high-concentration intravenous formulations. Collectively, PEG linkers function as multifunctional molecular tools. Through the synergistic effects of their shielding, solubilization, and controlled-release capabilities, they systematically overcome multiple obstacles—including recognition, clearance, and stability—that traditional drugs encounter during in vivo delivery.
PurePEG’s diverse range of PEGylation reagents offers multiple options for enhancing the sustained-release properties of pharmaceuticals.
Benefits of Using PEG Linkers in Controlled Drug Release
Enhancing therapeutic efficacy
PEG linkers significantly improve the bioavailability of therapeutic agents by enhancing drug accumulation in tumors and prolonging retention time at the lesion site. This effect is particularly crucial for therapies requiring sustained drug exposure—for instance, in chemotherapy, maintaining effective intracellular drug concentrations is a key strategy to overcome tumor cell resistance mechanisms; in chronic disease treatment, stable drug levels ensure continuous suppression of pathological processes. By precisely controlling PEG chain length and branching structure, drug distribution characteristics within target tissues can be further optimized, enabling targeted enhancement of therapeutic potency.
Extending systemic circulation time
PEG modification effectively delays renal filtration rates by increasing the drug’s hydrodynamic volume. Simultaneously, its hydrophilic shell reduces non-specific interactions with blood components and immune recognition. This improvement in pharmacokinetic parameters not only significantly extends the drug’s plasma half-life, allowing for optimized dosing intervals, but more importantly, ensures sustained therapeutic efficacy by maintaining stable blood drug concentrations. This provides a technical foundation for improving patient compliance and quality of life.
Reduced systemic toxicity
Through synergistic passive targeting (EPR effect) and active controlled-release mechanisms, PEG linkers deliver highly active therapeutic molecules specifically to diseased regions. This spatial distribution control minimizes unnecessary drug exposure to healthy tissues, thereby reducing dose-limiting toxicity—a common issue with traditional delivery methods—at its source. For potent drugs with narrow therapeutic windows, this precision delivery strategy enables significant improvements in drug safety and tolerability while maintaining or even enhancing anticancer activity. It represents a key technological pathway to overcome existing efficacy bottlenecks.
Applications of PEG Linkers in Medicine
Polyethylene glycol (PEG) linker technology, as a core component of modern drug delivery systems, demonstrates critical value across multiple cutting-edge therapeutic fields. Its application has evolved from simple molecular modification into an engineered platform for precision drug design.
Cancer therapies and targeted delivery
PEG linkers serve as critical bridges for constructing antibody-drug conjugates (ADCs) through their functionalizable terminal groups. They not only enable precise delivery of cytotoxic drugs to tumor sites but also maintain the stability of the entire conjugate during systemic circulation via their hydrophilicity and steric hindrance effects. This approach significantly enhances the therapeutic index while substantially reducing off-target toxicity, providing a vital technological pathway for tumor-targeted therapy.
Nano-drug design
PEGylation modifications form a dynamic hydration layer on carrier surfaces, effectively reducing protein adsorption and immune cell recognition. This significantly enhances the stealth capability and prolonged circulation properties of delivery systems like nanoparticles and liposomes. This “stealth effect,” combined with the high permeability and retention (EPR) effect characteristic of solid tumors, promotes selective drug accumulation at the tumor site, offering an effective strategy to overcome the tissue distribution limitations of traditional chemotherapeutic drugs.
PurePEG’s PEG45 (high purity, monodisperse), PEG-Lipid, RROTAC, and other products are highly suitable for these advanced formulations.
Therapeutic proteins and peptide drugs
The introduction of PEG linkers significantly improves the pharmacokinetic properties of these biologics by increasing their hydrodynamic volume and shielding protease cleavage sites. This covalent modification not only delays renal clearance and enzymatic inactivation but also creates a more favorable in vivo environment by reducing immunogenicity. This enables the development of long-acting formulations, extending dosing intervals from daily to weekly or even monthly, fundamentally reshaping the clinical utilization patterns of biologics.
Future of PEG Linkers in Controlled Drug Release
The field of PEG chemistry is continuously evolving, with researchers working to overcome current limitations and unlock new possibilities.
Innovations in PEG linker design
New linker designs are emerging that offer even greater control. This includes multi-component linkers that respond to dual triggers (e.g., pH and a specific enzyme) for highly specific drug release. Additionally, the development of precisely structured linkers, like our Multi-Arm PEGs, allows for the creation of more complex and functional drug conjugates.
Trends in personalized medicine and advanced drug delivery
As medicine becomes more personalized, drug delivery systems will need to become more adaptable. Future PEG linkers may be designed to respond to biomarkers unique to an individual patient’s disease, paving the way for truly personalized therapies.
Conclusion: Why PEG Linkers Matter in Controlled Drug Release
Polyethylene glycol (PEG) linkers have evolved from simple molecular spacers into core enabling technologies in drug delivery. By precisely regulating drug release kinetics, significantly enhancing solubility and circulation half-life, and effectively reducing systemic toxicity, they have successfully transformed numerous drug candidates into clinically viable therapeutic solutions. Although challenges remain in long-term stability and immunogenicity, precision preparation techniques—represented by monodisperse PEG—continue to drive breakthroughs in overcoming these bottlenecks. As a critical component of precision medicine systems, PEG linkers provide an indispensable technological foundation for the clinical translation of cutting-edge therapeutic modalities, from targeted anticancer therapies to smart nanomedicines. They continuously propel the evolution of drug design paradigms toward higher specificity and controllability.
At PurePEG, we are committed to providing high-purity, monodisperse PEG reagents to advance the application of PEG in nanomedicine. You may also contact us for customized product services to create research molecules tailored specifically for your needs.