Introduction: The Critical Role of PEG Linker Selection
The selection of an appropriate PEG linker is a pivotal step in developing antibody-drug conjugates (ADCs) and other bioconjugates, as it critically influences the drug’s stability, solubility, efficacy, and safety—far beyond merely acting as a spacer. A suboptimal choice can lead to experimental failure, poor in vivo performance, and development delays. Given the profound implications of linker selection on pharmacokinetics and payload delivery, this guide aims to outline common pitfalls and provide strategies for optimal selection.
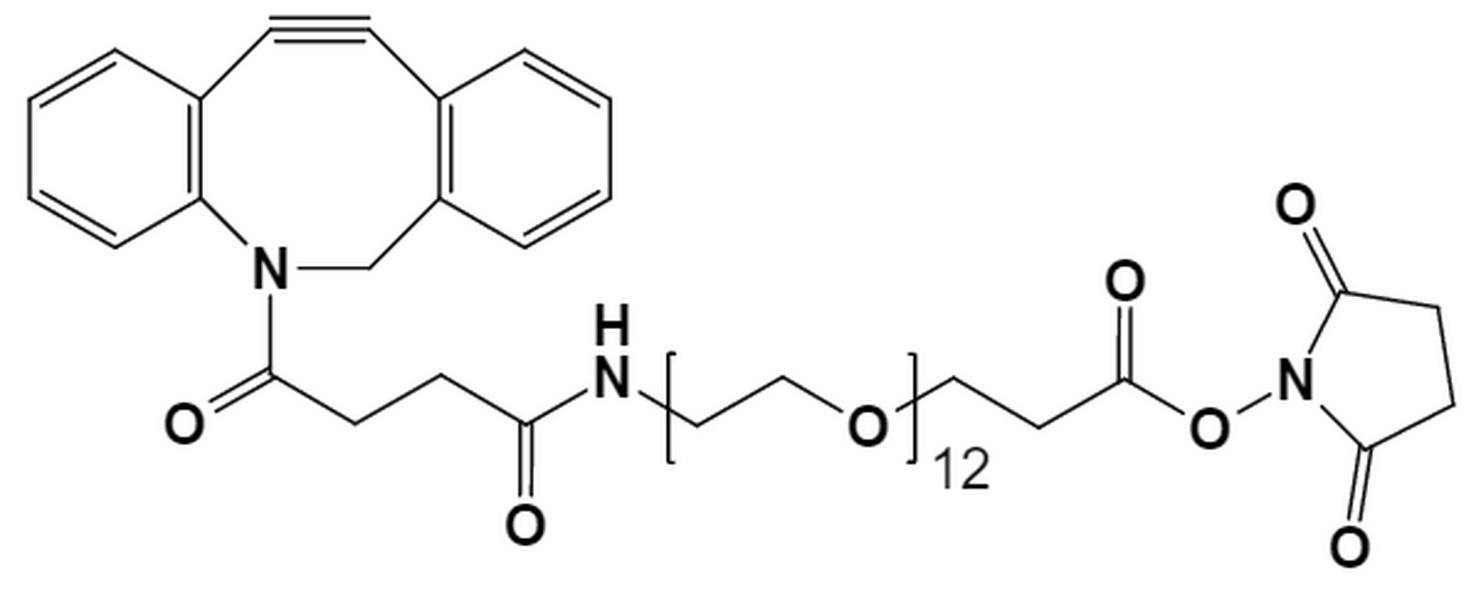
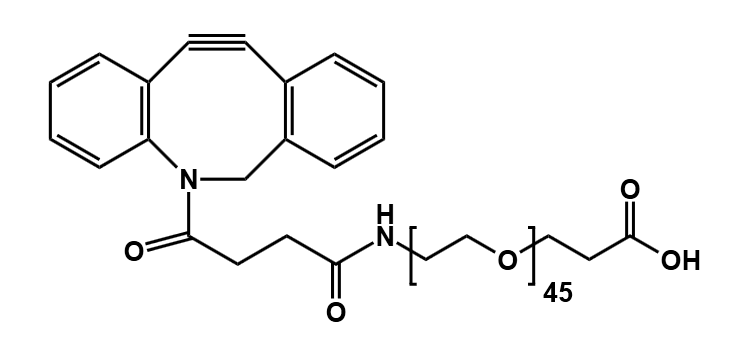
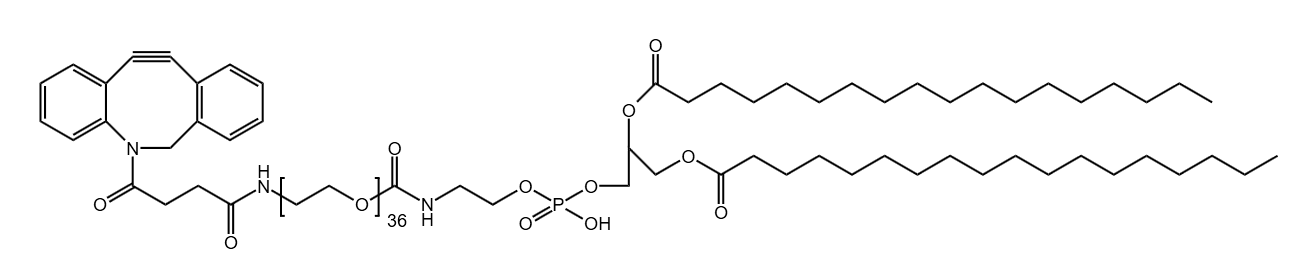
Pitfall 1: Selecting a Suboptimal PEG Molecular Weight
A frequent error is the selection of a PEG chain with an inappropriate length. The molecular weight, or chain length, of the PEG linker directly modulates the properties of the final conjugate.
Impact on Solubility and Pharmacokinetics
The PEG chain length significantly affects the conjugate’s hydrodynamic volume. Longer chains increase overall size, which can prevent rapid clearance by the kidneys and significantly extend the drug’s circulation half-life. However, excessive length may impede tissue penetration or hinder target binding.
Conversely, a chain that is too short may provide insufficient solubility for hydrophobic payloads, leading to aggregation, and may offer inadequate shielding from immune recognition, resulting in accelerated clearance. Identifying the optimal length is crucial for balancing solubility, pharmacokinetics, and biodistribution.
Strategies for Optimal PEG Length Selection
- Define Primary Objectives: Determine whether the goal is extended half-life or enhanced tissue penetration. For long-circulating applications, longer chains (e.g., from our PEG45 products) are often suitable.
- Evaluate Payload Hydrophobicity: Highly hydrophobic payloads typically require longer PEG chains to ensure solubility.
- Conduct Empirical Screening: Test a limited range of discrete PEG lengths (e.g., PEG8, PEG12, PEG24) to compare conjugate performance in relevant assays.
- Utilize Monodisperse PEGs: Using monodisperse PEGs from PurePEG ensures that every conjugate has the exact same chain length, eliminating the variability that plagues polydisperse products.
Pitfall 2: Overlooking the Hydrophilicity-Hydrophobicity Balance
Failing to adequately balance the hydrophilic PEG chain with the hydrophobic drug payload can lead to significant formulation and delivery challenges..
Consequences on Solubility, Aggregation, and Delivery
Potent payloads are often highly hydrophobic. Without sufficient counterbalancing hydrophilicity from the linker, the conjugate may aggregate, compromising efficacy and increasing the risk of immunogenicity. Aggregated species are rapidly cleared by the reticuloendothelial system, reducing target exposure.
Guidelines for Achieving an Optimal Balance
- Quantify Payload Hydrophobicity: Use computational or experimental methods to assess logP or other hydrophobicity metrics.
- Select an Appropriately Long PEG Chain: Longer chains contribute greater hydrophilicity. For challenging payloads, consider our PEGylation Reagents.
- Incorporate Hydrophilic Spacers: Besides the main PEG chain, some linkers include other hydrophilic components to further improve water solubility.
- Optimize Formulation: Even with an optimal linker, the final formulation conditions (pH, buffers, excipients) are critical for maintaining stability and preventing aggregation.
Caption: A hydrophilic PEG linker creates a protective water-soluble shield around a hydrophobic payload, preventing aggregation and improving stability.
Featured Products
Pitfall 3: Misjudging Cleavable vs. Non-Cleavable Linkers
The choice between a cleavable and a non-cleavable linker dictates the payload release mechanism and must align with the therapeutic’s intended mechanism of action.
Impact on Drug Release and Therapeutic Index
- Cleavable Linkers: These are designed to release the payload upon encountering a specific trigger inside the target cell (e.g., low pH in endosomes or specific enzymes like cathepsins). This allows for targeted intracellular drug release, maximizing potency against cancer cells while minimizing systemic exposure. Our Cleavable Linkers are engineered for this precise control.
- Non-Cleavable Linkers: These form a permanent bond. The drug is only released when the entire antibody is degraded within the cell’s lysosome. This can be advantageous for payloads that are still active when attached to a single amino acid, but it relies entirely on antibody catabolism.
An improperly selected cleavable linker with low plasma stability can cause premature release and dose-limiting toxicity. Conversely, a non-cleavable linker used with a payload requiring free activity will yield an inactive conjugate.
Selecting the Appropriate Linker Type
- Understand Your Payload’s MOA: Does the drug need to be completely free to bind its intracellular target? If yes, a cleavable linker is almost always necessary. A successful cleavable PEG linker synthesis strategy often involves enzyme-sensitive sequences like Val-Cit, found in products like Fmoc-PEG4-Val-Cit-PAB-OH.
- Analyze the Target Environment: Is there a unique feature of the tumor microenvironment you can exploit? For example, the hypoxic core of some tumors can be targeted with hypoxia-sensitive linkers.
- Evaluate the “Bystander Effect”: If the payload is released and is membrane-permeable, it can exit the target cell and kill nearby antigen-negative cancer cells. This “bystander effect” is only possible with cleavable linkers.
Pitfall 4: Underestimating Stability and Degradation Risks
Linker instability, whether chemical or physical, can compromise the entire conjugate. Ensuring robustness is paramount.
Challenges in Chemical and Physical Stability
Chemical instability can lead to premature cleavage in the bloodstream, while physical instability can cause aggregation. For example, linkers with maleimide groups for thiol conjugation can undergo a retro-Michael reaction, leading to deconjugation. Some ester bonds are susceptible to hydrolysis.
Physical instability often stems from the issues of hydrophobicity discussed earlier. Ensuring the final conjugate remains a stable, soluble monomer is a primary goal of ADC development.
Strategies for Enhancing Linker Robustness
- Employ Stable Conjugation Chemistries: For thiol conjugation, consider next-generation maleimide derivatives or other chemistries that form more permanent bonds.
- Optimize Storage and Formulation Conditions: Store and formulate the ADC at a pH that minimizes hydrolysis of sensitive bonds.
- Source High-Purity Linkers: Impurities in linker preparations can introduce reactive side groups that compromise stability. PurePEG’s >99% pure, monodisperse linkers, such as our Heterobifunctional PEGs, provide the consistency needed for a robust final product.
- Conduct Comprehensive Stress Testing: Expose your ADC to various conditions (e.g., different temperatures, pH levels) to identify potential degradation pathways early in the development process.
Pitfall 5: Neglecting Manufacturing and Scalability
A linker that performs well at the research scale may be unsuitable for GMP manufacturing. Scalability must be considered from the outset.
Challenges in Batch Consistency and GMP Compliance
Polydisperse PEGs introduce inherent heterogeneity, complicating quality control and regulatory approval. Complex synthetic routes can yield low amounts and introduce difficult-to-remove impurities.
Planning for Scale-Up
- Prioritize Monodisperse PEGs: Start with a monodisperse PEG linker for ADCs. This is the single most important factor for ensuring a homogeneous, reproducible product.
- Choose Efficient Conjugation Chemistries: Where possible, opt for linkers that can be synthesized in high yields with straightforward purification steps.Such as our Clickable Linkers.
- Engage with Your Supplier Early: Discuss your scalability needs with your linker provider. At PurePEG, we offer custom synthesis and can advise on which of our catalog products are best suited for large-scale production.
CTA: Talk to a PurePEG scientist for guidance
Navigating these pitfalls requires expertise. The optimal choice is rarely obvious and often involves balancing multiple competing factors. If you are struggling with linker selection or troubleshooting an existing conjugate, our team of expert scientists is here to help.
Don’t let linker selection derail your project. Contact a PurePEG expert today for a free consultation and let us help you find the perfect molecular tool for your groundbreaking research.