Antibody-drug conjugates (ADCs) represent a transformative class of targeted cancer therapeutics, combining the specificity of monoclonal antibodies with the potent cytotoxicity of small-molecule payloads. The linker in its structure plays a crucial regulatory role in the stability, solubility, and payload release of the ADC.
Introduction to Linker Chemistry in ADCs
Overview of Antibody-Drug Conjugates (ADCs)
Antibody-drug conjugates (ADCs) are rationally designed targeted biopharmaceuticals integrating three distinct functional modules: a monoclonal antibody specifically targeting tumor antigens, a highly cytotoxic payload, and a chemical linker covalently connecting the two. The fundamental objective of this design is to achieve specific accumulation and release of cytotoxic drugs at tumor sites. This maximizes antitumor efficacy while minimizing systemic exposure and adverse effects on normal tissues.
The Critical Role of Linker Chemistry in ADC Efficacy
Within the ADC architecture, the linker is far from an inert connecting unit; it serves as the critical functional hub directly regulating the therapeutic window. Its chemical properties must achieve a delicate balancing act: maintaining exceptional stability in the systemic circulation to prevent premature leakage of the payload during delivery, thereby avoiding off-target toxicity; simultaneously, upon ADC internalization by tumor cells, the linker must respond to specific intracellular signals (such as lysosomal proteases or low pH environments) to be efficiently activated and release the active payload. This dual requirement for circulatory stability and efficient intracellular release constitutes the fundamental challenge in ADC drug design, making linker chemistry the core R&D focus driving the entire field forward.
Featured Products
Impact of Linker Chemistry on ADC Stability
The chemical design of linkers is pivotal in determining the efficacy and safety of antibody-drug conjugates (ADCs). Their stability encompasses both chemical and physical dimensions: chemical stability ensures the integrity of covalent bonds between the linker and payload during circulation, preventing off-target toxicity; physical stability concerns the macroscopic state of the entire conjugate, preventing molecular aggregation caused by hydrophobic or electrostatic interactions that could compromise pharmacokinetics and immunogenicity.
Modern ADC development focuses on rationally designing the covalent bond type and overall molecular conformation of linkers to achieve high stability in plasma. Take enzyme-cleavable linkers (e.g., the classic MC-Val-Cit-PAB-OH) as an example: they exhibit excellent chemical inertness in blood but are specifically recognized and cleaved by highly active proteases (such as cathepsin B) within tumor cells after internalization and transport to lysosomes, enabling precise intracellular drug release.
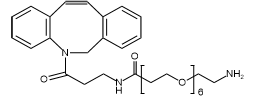
Furthermore, the application of bioorthogonal chemistry enhances the robustness of conjugation techniques. For instance, strain-promoted azide-alkyne cycloaddition (SPAAC) efficiently forms triazole bonds that remain exceptionally stable under physiological conditions. When combined with hydrophilic modules like polyethylene glycol (PEG), such techniques not only significantly improve the solubility of conjugates but also effectively suppress their chemical degradation and physical aggregation. This provides a robust technical foundation for preparing structurally uniform and stable ADC products.
How Linker Chemistry Influences Solubility
In the design of antibody-drug conjugates (ADCs), the chemical properties of the linker serve as a key variable in regulating the overall solubility of the conjugate. Linkers can be rationally designed based on their hydrophilic-hydrophobic properties: selecting hydrophilic linkers is one of the core strategies for addressing solubility challenges posed by strongly hydrophobic cytotoxins. Insufficient solubility not only triggers ADC aggregation, leading to loss of efficacy, but may also induce immunogenic responses. Furthermore, it presents significant formulation development challenges, such as necessitating the use of irritant excipients or preventing attainment of effective therapeutic concentrations.
Polyethylene glycol (PEG)-based linkers exemplify this strategy. By adjusting PEG chain length and structure, the hydrophilic-hydrophobic balance of ADCs can be precisely modulated. For instance, employing monodisperse PEG45 reagents effectively streamlines the development of stable, high-concentration injectable formulations, ensuring adequate bioavailability following intravenous administration.
Linker Chemistry and Controlled Drug Release
Cleavable vs. Non-Cleavable Linkers Strategies
Linker chemistry is central to regulating the intracellular release behaviour of antibody-drug conjugates (ADCs), with its design hinging on achieving a precise equilibrium between circulatory stability and target-specific release. Based on differing release mechanisms, linkers primarily fall into two strategies: cleavable and non-cleavable. This fundamental choice directly determines the final form and mode of action of the active payload.
Cleavable linkers achieve targeted activation by responding to specific biological signals within the tumour cell-specific microenvironment. Their primary mechanisms include:
- Enzymatic cleavage: Utilising proteases highly expressed in lysosomes (e.g., cathepsin B) to specifically cleave dipeptide sequences such as valine-citrulline (Val-Cit), enabling efficient intracellular drug release.
- Acid-sensitive cleavage: Leveraging the acidic environment (pH 4.5–0) of endosomes/lysosomes, chemical bonds such as pyridine bonds undergo selective hydrolysis intracellularly while remaining stable in neutral blood plasma.
- Reduction-sensitive cleavage: Utilising high intracellular glutathione concentrations to specifically cleave disulphide bonds, enabling rapid cytoplasmic release.
In contrast, non-cleavable linkers rely on complete degradation of the antibody chain within lysosomes, ultimately releasing a payload derivative still bound to amino acid residues. Whilst this strategy offers exceptional plasma stability, it necessitates that the resulting metabolite retains sufficient cytotoxic activity.
Balancing Stability with Targeted Release
Ideal linker design must strike a balance between ‘sufficient stability’ and ‘sufficient sensitivity’. Excessive stability compromises therapeutic efficacy, while premature cleavage triggers off-target toxicity. Consequently, precisely engineering linker chemistry to maximise the therapeutic window of ADCs represents a central objective and challenge in contemporary drug design.
PEG Linker: A Key Module in ADC Linker Chemistry
Definition and Types of PEG Linkers
PEG linkers are indispensable functional modules in the design of antibody-drug conjugates (ADCs). Essentially linear polymers composed of repeating ethylene oxide units, they play a central role in regulating the physicochemical properties of ADCs and facilitating their in vivo delivery due to their exceptional hydrophilicity and molecular chain flexibility. PurePEG provides high-purity, monodisperse PEGs, which are singular molecular weight entities. This homogeneity is crucial for producing consistent ADCs with predictable pharmacokinetics and safety profiles.
PEG Linkers: From Enhanced Pharmacokinetics to Precise Targeted Release
The functionality of PEG linkers is primarily manifested in the systematic optimization of ADC drug properties. Many highly active cytotoxic payloads exhibit strong hydrophobicity, readily inducing ADC aggregation in aqueous environments and consequently compromising drug stability and efficacy. The PEG chains form a “hydration shell” around the molecule through their potent hydration effects, significantly enhancing the solubility of the entire ADC. Simultaneously, this structure increases the drug’s hydrodynamic volume, effectively slowing renal clearance rates and prolonging its plasma half-life. This creates favorable conditions for effective drug accumulation within tumor tissues.
Furthermore, PEG linkers enhance ADC systemic stability through a spatial shielding effect. Its flexible hydrophilic chains form a dynamic protective layer on the ADC surface, reducing non-specific interactions with blood components. This decreases protein aggregation tendencies, weakens immunogenic recognition, and slows capture and clearance by the hepatic reticuloendothelial system. This mechanism helps maintain ADC structural integrity during circulation, ensuring more active drug reaches target cells with precision.
To achieve these functions, PEG linkers can undergo diverse molecular designs.
Common types include:
- Heterobifunctional PEGs: Featuring different reactive groups on each terminus, they enable controlled, sequential conjugation of the antibody and payload, making them ideal for constructing well-defined ADCs.
- Cleavable Linkers: Engineered with labile bonds that break under specific intracellular conditions.
- Clickable Linkers: Incorporate bioorthogonal functional groups (e.g., DBCO, Azide) for highly efficient and specific conjugation under mild conditions.
To optimise the solubility of PEG, the following methods may be employed.
- Increase PEG Chain Length: Using linkers with more ethylene glycol units (e.g., PEG12, PEG24, or PEG45) directly increases hydrophilicity.
- Incorporate Charged Groups: Linkers with charged or polar groups can further enhance water solubility.
- Utilize Branched Architectures: Multi-Arm PEGs create a dense hydrophilic shield that is highly effective at solubilizing challenging payloads.
Factors to be considered by the R&D team when selecting PEG linkers
- Payload Characteristics: Hydrophobicity, potency, and requirement for release in its native form.
- Target Antigen Biology: Internalization rate and enzymatic profile of the target cell.
- Conjugation Strategy: Available functional groups on the antibody/payload and desired site-specificity.
- Desired Pharmacokinetic Profile: Target circulation half-life.
An Excellent Supplier of Monodisperse PEG Linkers
PurePEG has specialised in monodisperse PEGs for many years, with our suppliers possessing mature production capabilities and cutting-edge technical expertise. Furthermore, we have supported many research institutions in advancing novel drug molecule development.
We offer a diverse range of monodisperse PEG products for researchers to select from, including PEG45, heterobifunctional PEGs, and homobifunctional PEGs. These are suitable for applications across multiple fields, such as Protein cross-linking, peptide cross-linking, targeted drug delivery, and PROTAC development.
Explore our full range of PEG products or contact our experts to discuss your custom synthesis needs.