Introduction: The Role of Smart Linkers in Targeted Therapies
In advanced drug delivery, such as Antibody-Drug Conjugates (ADCs) and nanoparticle-based delivery systems, the linker is a critical component that dictates therapeutic success. “Smart” linkers are sophisticated molecular bridges designed to remain stable in circulation and to release their drug payload only in response to specific environmental triggers within the target tissue or cell. This controlled release mechanism maximizes therapeutic efficacy at the disease site while minimizing off-target toxicity to healthy tissues.
This document provides an overview of the primary stimuli-responsive linker technologies, detailing their mechanisms, triggers, and ideal applications in modern medicine.
1. pH-Sensitive Linkers
Mechanism: These linkers incorporate acid-labile functional groups, such as hydrazones or cis-aconityl bonds, that are stable at neutral pH (blood pH ≈ 7.4) but undergo rapid hydrolysis (and subsequent cleavage) in acidic environments.
Trigger: Low pH (typically pH 4.5-6.5).
Biological Context:
- Tumor Microenvironment: The extracellular space around solid tumors is often slightly acidic.
- Cellular Endosomes & Lysosomes: After a therapeutic is internalized by a cell, it is trafficked into endosomes and lysosomes, where their internal pH drops significantly (pH 4.5-5.5). This is the most common and reliable trigger for pH-sensitive cleavage.
Applications:
- Targeting solid tumors.
- Intracellular release of cytotoxic agents from ADCs.
- Delivery of nucleic acids via nanoparticle carriers.
2. Enzyme-Cleavable Linkers
Mechanism: These linkers contain a specific peptide sequence or other motifs that are recognized and cleaved by enzymes overexpressed at the disease site.
Trigger: Presence of a specific enzyme.
Biological Context:
- Cancer Cells: Many tumors overexpress certain proteases, such as cathepsins (e.g., Cathepsin B), matrix metalloproteinases (MMPs), or plasmin.
- Inflammatory Sites: Areas of inflammation often have high concentrations of specific enzymes.
Common Examples:
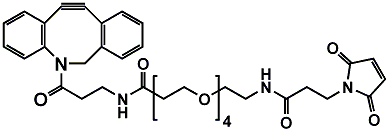
- Valine-Citrulline (Val-Cit): A dipeptide sequence efficiently cleaved by Cathepsin B inside lysosomes—making it one of the most clinically validated linkers for ADCs.Pure PEG can offer a variety of Val-Cit products, such as: Mal-PEG2-Val-Cit-PAB-OH, Azido-PEG2-Val-Cit-PAB-Exatecan.
- Valine-Alanine (Val-Ala): Another peptide sequence used for enzyme-targeted cleavage.Pure PEG can offer a variety of Val-Ala products, such as: Fmoc-PEG4-Val-Ala-PAB, Mal-PEG8-amide-Val-Ala-Exatecan.
Applications:
- Gold standard for many FDA-approved and clinical-stage ADCs.
- Highly specific drug release within tumor cells, minimizing the bystander effect until cleavage.
Featured Products
3. Redox-Sensitive Linkers
Mechanism: These linkers feature a disulfide bond (-S-S-), stable in the oxidizing environment of the bloodstream but rapidly reduced and cleaved in the intracellular reducing environment. PurePEG offers two main product categories: Disulfide Linkers and Disulfide PEG Linkers, including various products such as DBCO-PEG4-S-S-PEG4-DBCO, Azidoethyl-SS-amido-PEG9-acid, and Boc-NH-ethyl-SS-propionic acid.
Trigger: High concentration of intracellular reducing agents, primarily glutathione (GSH).
Biological Context:
- Intracellular Environment: The concentration of glutathione inside a cell (~1-10 mM) is significantly higher than in the extracellular space or blood plasma (~1-10 µM). This creates a steep redox gradient.
- Tumor Cells: Many cancer cells exhibit elevated GSH levels due to oxidative stress, amplifying the redox trigger specificity.
Applications:
- Intracellular delivery of small molecule drugs, siRNAs, and other therapeutic payloads.
- Redox-responsive nanoparticles that disassemble upon GSH-mediated cleavage, releasing cargo post-internalization.
4. Photocleavable Linkers
Mechanism: These linkers contain a light-labile group—typically a nitrobenzyl moiety—stable until irradiation with light of a specific wavelength (typically UV or near-infrared). The light provides the energy to irreversibly cleave the linker and release the payload.
Trigger: External light source of a specific wavelength.
Biological Context:
- This is an externally controlled mechanism, not dependent on endogenous biological conditions. The trigger is applied by a clinician or researcher.
Applications:
- Spatially-Controlled Release: Ideal for treating localized, surface-level diseases like skin cancers or certain ocular conditions where light can be precisely directed.
- Research Tools: Used in “optogenetics” and cell biology to study cellular processes with high temporal and spatial precision.
- Photodynamic Therapy (PDT): Can be used to release cytotoxic agents or photosensitizers exactly at the target site.