The rapid development of mRNA vaccines has marked a turning point in modern medicine, offering a powerful new platform to combat infectious diseases. Behind this breakthrough lies a sophisticated delivery system, often driven by lipid nanoparticles (LNPs). A critical component of these LNPs is the Polyethylene Glycol (PEG) linker, a molecular tool that provides stability, enhances delivery, and minimizes unwanted immune reactions.
This article explores the essential PEG linker strategies used in mRNA vaccine development. We will break down what PEG linkers are, how they contribute to building effective LNPs, the benefits they offer, and the challenges that researchers face.
What Are PEG Linkers?
Polyethylene Glycol (PEG) linkers are biocompatible polymers composed of repeating ethylene glycol units, featuring reactive functional groups at both ends that serve as molecular bridges to connect components in complex formulations such as lipid-based mRNA vaccine delivery systems. Known for their non-toxic, non-immunogenic, and highly flexible nature, PEG linkers form a protective hydrophilic shield around nanoparticles, preventing aggregation and premature immune clearance. By enhancing the solubility and stability of hydrophobic drug molecules or carriers through PEGylation, these linkers not only improve biocompatibility but also extend circulation time by increasing nanoparticle size to reduce renal filtration—thereby maximizing targeted delivery efficiency and therapeutic response.
At PurePEG, we provide high-purity, monodisperse PEGs to ensure uniformity and predictable performance in these critical applications.
PEG Linker Strategies in mRNA Vaccine Design
PEGylation for lipid nanoparticles (LNPs)
Lipid nanoparticles are the primary delivery systems for mRNA vaccines. They are tiny spheres made of lipids that encapsulate and protect the fragile mRNA molecules. A key ingredient in this lipid mixture is a PEG-lipid conjugate. This consists of a lipid “anchor” that embeds into the LNP surface and a PEG chain that extends outward.
Our portfolio includes a variety of PEG-Lipid products designed for this purpose. For example, molecules like mPEG36-DSPE and mPEG44-DSPE are frequently used to form the protective outer layer of LNPs, ensuring the vaccine remains stable until it can be delivered to cells.
Optimizing linker length and hydrophilicity
The length of the PEG chain is a critical design parameter. A longer PEG chain provides a thicker protective layer, offering greater stability and longer circulation. However, a chain that is too long can sometimes hinder the nanoparticle’s ability to fuse with the cell membrane and release its mRNA payload, a phenomenon known as the “PEG dilemma.”
Researchers must carefully balance linker length to optimize both stability and delivery efficiency. PurePEG offers a wide range of chain lengths, from short PEG chains to longer polymers like monodisperse PEG45 products, allowing scientists to fine-tune their LNP formulations for maximum effectiveness.
Functionalization for targeted delivery
While current mRNA vaccines are designed for general distribution, future strategies may involve targeting specific cell types to enhance the immune response or develop therapeutic vaccines. This can be achieved by using Heterobifunctional PEGs to attach targeting ligands (such as antibodies or peptides) to the LNPs’ surface.
For example, a linker with a DBCO group, like DBCO-CONH-PEG45-DSPE, can be used with click chemistry to attach an azide-modified targeting molecule. This approach, often used to create a PEG linker for ADCs, can be adapted for vaccines to guide LNPs to specific immune cells.
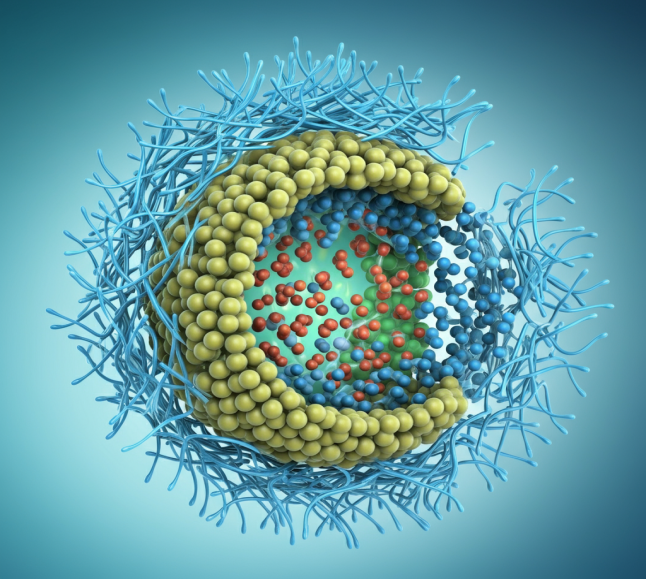
Benefits of PEG Linkers in mRNA Vaccines
Improved stability and shelf-life
The PEG layer prevents LNPs from aggregating during storage, which is crucial for maintaining a consistent particle size and ensuring dose accuracy. This steric stabilization significantly improves the vaccine’s shelf-life, allowing it to be transported and stored until it is ready for administration.
Reduced immunogenicity and adverse reactions
By shielding the LNP from the immune system, PEGylation reduces the chance that the delivery vehicle itself will be recognized as a foreign invader and cleared prematurely. This “stealth” effect helps minimize inflammatory responses and ensures the mRNA has enough time to be translated into the target antigen, leading to a focused and effective immune response.
Enhanced delivery efficiency and uptake
The PEG shield plays a delicate role in cellular uptake. While it provides stability in circulation, the PEG-lipid must detach or rearrange at the cell surface to allow the LNP to fuse with the cell membrane. The precise density and length of the PEG chains are optimized to balance this process, ensuring efficient delivery of the mRNA payload into the cell’s cytoplasm. Our DMG-PEG products are an example of specialized lipids used to fine-tune this balance.
Challenges and Considerations
Potential PEG-related immune responses
Although rare, some individuals have pre-existing anti-PEG antibodies from prior exposure to PEG in cosmetics, foods, or medicines. These antibodies can, in some cases, lead to rapid clearance of the LNPs or, even more rarely, cause allergic reactions. Research is ongoing to understand and mitigate this potential immunogenicity, including the development of next-generation linkers.
Optimization for dose and pharmacokinetics
Determining the optimal PEG density and length is a complex process. Too much PEG can reduce cellular uptake, while too little can lead to instability and rapid clearance. This requires careful screening and optimization studies to find the right pharmacokinetic profile for each specific vaccine. The use of high-purity, monodisperse PEGs, where every molecule has the same chain length, is critical for achieving reproducible results in these studies.
Regulatory and manufacturing challenges
Manufacturing PEG-lipids is a complex process that requires strict quality control. Any impurities or variations in PEG chain length (polydispersity) can impact the vaccine’s safety and efficacy. Regulatory agencies demand extensive characterization of all components.
PurePEG is your trusted partner, offering high-purity, monodisperse PEGs across multiple categories and polymerisation degrees. We provide detailed certificates of analysis and support the custom synthesis of cleavable PEG linkers, enabling you to conduct cutting-edge research in this field.
Case Studies: PEG Linkers in COVID-19 mRNA Vaccines
Overview of successful PEGylated LNPs in approved vaccines
The two pioneering mRNA vaccines for COVID-19 have both utilized PEG-lipid conjugates in their LNP formulations. These vaccines demonstrated the power of PEGylation to create stable, effective, and safe delivery systems on a global scale. The specific PEG-lipid used in these formulations provides the stealth characteristics that allows the LNPs to circulate long enough to be taken up by cells, where the mRNA is then translated to produce the viral spike protein antigen.
Lessons learned for future vaccine development
The success of the COVID-19 vaccines provided a wealth of data on the performance of PEGylated LNPs in humans. It confirmed that a well-designed PEG-lipid is essential for stability and efficacy. It also highlighted the need for continued research into potential anti-PEG immune responses and spurred innovation in next-generation delivery platforms to further improve safety and performance.
Future Directions and Innovations
Next-generation PEG linkers and alternatives
To address the potential for anti-PEG immunogenicity, researchers are developing novel linker technologies. This includes creating biodegradable linkers that are cleaved after administration or exploring alternative hydrophilic polymers. PurePEG is at the forefront of this research, offering custom synthesis services to develop novel linkers, including Biotinylation Reagents and PROTAC linkers that can be adapted for new delivery systems.
Strategies to further improve mRNA vaccine delivery
Innovation is not limited to linkers. New lipid chemistries and LNP structures are being developed to improve targeting, enhance endosomal escape (the process of getting the mRNA out of the endosomal compartment and into the cytoplasm), and reduce the required dose. Combining these advanced lipids with precisely engineered PEG linkers will pave the way for even more effective vaccines.
Personalized and precision vaccine approaches
The flexibility of the mRNA platform opens the door to personalized vaccines, such as those tailored to a patient’s specific cancer mutations. In these applications, targeted delivery will be essential. This will require sophisticated linker strategies, similar to those used in the best PEG linker for protein conjugation, to attach targeting moieties to the LNP surface and direct the vaccine to the intended cells.

