Executive Summary
A biopharmaceutical partner encountered a critical development hurdle with a promising Antibody-Drug Conjugate (ADC). The candidate molecule, incorporating a highly potent yet extremely hydrophobic payload, exhibited severe aggregation and poor solubility, rendering it unsuitable for clinical formulation. By replacing the original non-PEG linker with PurePEG’s monodisperse Mal-PEG8-COOH, the partner achieved a 10-fold increase in solubility and a 6-fold reduction in aggregation after seven days in serum. This intervention transformed a non-viable candidate into a stable, manufacturable, and clinically promising therapeutic, demonstrating the critical role of high-purity PEG linkers in advanced ADC design.
Challenge: Overcoming Payload-Induced Aggregation in a Promising ADC
The client, a leading biopharmaceutical company, was developing a next-generation ADC for an oncology indication. The design utilized a well-characterized monoclonal antibody conjugated to a novel, highly potent cytotoxic payload. While early in vitro studies showed exceptional cell-killing activity, the program stalled during pre-formulation and development stages.
The primary challenges were:
- Poor Solubility: The ADC could not be concentrated beyond 2 mg/mL in standard phosphate-buffered saline (PBS) solution without visible precipitation. This was far below the typical clinical formulation requirement of >20 mg/mL.
- Significant Aggregation: Size Exclusion Chromatography (SEC) analysis revealed that over 30% of the ADC molecules existed as high-molecular-weight aggregates immediately after preparation. This level of aggregation is unacceptabledue to potential impacts on efficacy and immunogenicity risk.
- Manufacturing Challenges: The propensity for aggregation complicated downstream processing, including purification and sterile filtration, leading to high product loss and batch-to-batch variability.
The root cause was identified as the extreme hydrophobicity of the payload. The short, non-PEGylated linker in the initial design failed to provide adequate shielding, resulting in hydrophobic patches on the conjugate surface that drove intermolecular aggregation.
Solution: Rational Linker Re-Design with a Monodisperse PEG Spacer
PurePEG’s technical team collaborated with the client to re-engineer the linker architecture. The strategy was to incorporate a hydrophilic spacer to mitigate the payload’s hydrophobicity.
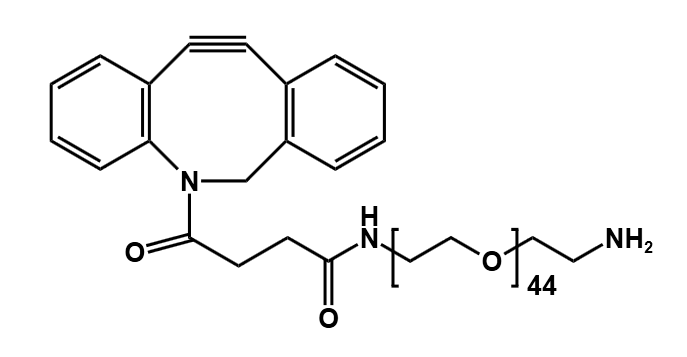
The selected solution was a discrete, heterobifunctional PEG linker from PurePEG: Mal-PEG8-COOH, with a certified purity of >99%.
Rationale for the Solution:
- Enhanced Hydrophilicity: The eight ethylene oxide units form a flexible, highly hydrous shell around the conjugated payload, significantly improving aqueous solubility.
- Monodispersity: Unlike polydisperse PEGs, this single-molecular-weight linker ensures structural homogeneity across the entire ADC population, simplifying analytical characterization and de-risking regulatory CMC requirements.
- Optimal Length: The PEG8 spacer provides sufficient length to prevent undesirable hydrophobic interactions without incurring potential pharmacokinetic complications associated with longer PEG chains.
- Orthogonal Conjugation Chemistry: The maleimide group allows for efficient, site-specific conjugation to engineered cysteine residues on the antibody, while the terminal carboxylic acid facilitates stable amide bond formation with the payload amine, enabling a controlled conjugation process.
Featured Products
Results: A 10-Fold Improvement in ADC Developability
The re-engineered ADC demonstrated dramatic and quantifiable improvements across all key metrics, validating the PEG linker strategy.
- Significantly Enhanced Solubility
The PEGylated ADC was successfully concentrated to over 20 mg/mL without any signs of precipitation or cloudiness. This represented a 10-fold increase in solubility, meeting the requirements for a clinical-grade intravenous formulation.
ADC Solubility Comparison
| Linker Type | Maximum Concentration (mg/mL) |
| Non-PEG Linker (Original) | 2 mg/mL |
| Mal-PEG8-COOH (PurePEG) | >20 mg/mL |
- Superior Stability Profile
Stability studies conducted in human serum at 37°C confirmed the enhanced stability conferred by the PEG linker.
- Reduced Aggregation: After 7 days, the PEGylated ADC showed less than 5% aggregation, compared to over 30% for the original ADC. This 6-fold reduction in aggregate formation points to a significantly more stable and safer product.
- Improved Linker Stability: The amount of free payload released into the serum was reduced from 15% (non-PEG linker) to less than 2% with the PEG linker, indicating a more robust chemical linkage.
Conclusion: High-Purity Monodisperse PEG Linkers as Essential Tools for ADC Development
This case study illustrates that rational linker design is a cornerstone of successful ADC development. The strategic incorporation of PurePEG’s high-purity monodisperse Mal-PEG8-COOH linker directly addressed the critical developability challenges posed by a hydrophobic payload. The result was a transformation in the candidate’s profile, achieving the solubility, stability, and homogeneity required for clinical advancement.
This success highlights the value of PurePEG’s proprietary technology, which produces precisely defined, monodisperse PEGs that empower scientists to build better, safer, and more effective therapeutics.
Customer Quote
“The aggregation issues with our lead ADC candidate put the entire project at risk. Switching to PurePEG’s monodisperse PEG8 linker was a game-changer. The improvements in solubility and stability were immediate and dramatic. PurePEG’s product quality and technical support were instrumental in getting our program back on track.”
– Principal Scientist, Biopharmaceutical Partner
Ready to solve your own bioconjugation challenges?
Explore our comprehensive portfolio of high-purity PEG linkers or contact our experts to discuss your custom synthesis needs.