The Rise of Smart Molecular Bridges
In the evolving world of bioconjugation, diselenide linkers (Se-Se bond linkers) are emerging as revolutionary tools. These dynamic connectors harness selenium’s unique chemistry to create “intelligent bridges” that respond to biological cues—releasing drugs precisely where needed. Unlike conventional linkers, they offer dual-triggered activation, breaking down in both reductive environments (like tumor cells) and acidic zones (e.g., inflamed tissues).
How Diselenide Linkers Work: Precision in Action
Imagine a drug conjugate as a locked safe. Diselenide linkers act as a smart lock that only opens under specific biological conditions:
- Reductive Unlocking: Inside cancer cells, high glutathione levels break the Se-Se bond, releasing the drug payload.
- Acidic Backup: In weakly reductive areas (like some tumors), acidic pH accelerates bond cleavage.
This two-step targeting minimizes damage to healthy tissues—a critical advance for therapies like antibody-drug conjugates (ADCs).
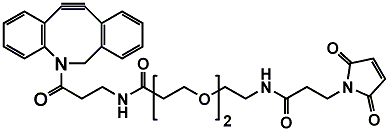
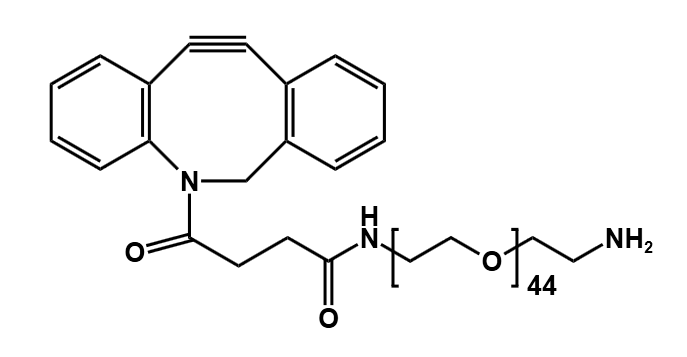
When paired with diselenide PEG, these linkers gain enhanced stealth and solubility. The PEG layer shields drugs from immune detection, while the diselenide core ensures tumor-specific activation.
Featured Products
Real-World Impact: From Labs to Clinics
Diselenide linkers are already reshaping medicine:
- Targeted Cancer Therapy: ADCs with diselenide linkers show superior tumor specificity and reduced liver toxicity compared to Val-Cit-based systems.
- mRNA Vaccine Delivery: They protect genetic cargo in blood and release it inside cells via reductive triggers.
- Smart Biomaterials: Wound dressings with diselenide bonds auto-release antibiotics in infected tissues.
Final Thoughts
Diselenide linker chemistry is expanding the frontiers of pharmaceutical innovation. By combining the precision of redox/pH-responsive cleavage with PEG’s pharmacokinetic advantages, these molecular tools enable unprecedented control over drug delivery. Whether advancing targeted cancer treatments, next-generation vaccines, or responsive biomaterials, diselenide linkers exemplify how smart chemistry drives real-world impact.
Actionable Insights:
- For targeted drug delivery (e.g., ADCs), prioritize diselenide linkers to minimize off-target effects.
- In mRNA/gene therapy, leverage diselenide-PEG hybrids to balance stability and intracellular release.
- Explore diselenide-based biomaterials for applications requiring stimuli-responsive release (e.g., infection management).
By mastering diselenide linker design, researchers can bridge the gap between laboratory promise and clinical reality.