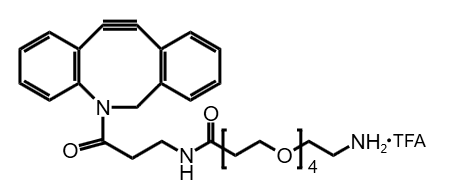
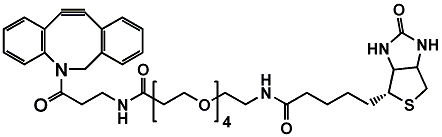
Poly(ethylene glycol), or PEG, remains one of the most versatile tools in modern bioconjugation and drug delivery. Within PEGylation strategies, the choice of functional groups can dictate whether a conjugation reaction is fast, selective, and reproducible — or frustratingly inefficient. Among these chemistries, maleimide-functionalized PEG linkers, especially PEG45, are among the most widely used options for thiol-specific conjugation.
In this deep-dive, we’ll cover:
- The underlying PEG45–maleimide chemistry and why it’s so selective for thiols.
- The advantages of maleimide linkers for protein, peptide, and antibody conjugation.
- Applications in thiol-based bioconjugation, including targeted drug delivery.
- The performance benefits of PEG45 as an intermediate-size spacer.
- Troubleshooting tips to overcome common PEGylation challenges.
Products referenced throughout this blog are available at PurePEG, we specialise in high-purity PEG linkers, including PEG45-Maleimide in various end-capped formats.
PEG45 Maleimide Chemistry Explained
PEG45 refers to a polyethylene glycol chain of approximately 45 ethylene oxide units (around 2 kDa molecular weight). This size offers an excellent balance — long enough to provide hydrophilic spacing between a payload and its carrier, but not so large as to overly increase molecular weight or interfere with biological function.
Maleimide chemistry is well-known for its high selectivity toward sulfhydryl groups (-SH). When a PEG linker is functionalized with a maleimide group, it can covalently react with cysteine residues on peptides, proteins, or other thiolated molecules under mild conditions.
- Reaction specificity: Maleimide groups form stable thioether bonds with thiols at physiological pH (~6.5–7.5), minimizing side reactions.
- Reaction kinetics: The maleimide–thiol reaction is fast, usually completing within minutes to hours depending on concentration and temperature.
- Stability of bond: The thioether linkage is non-reversible under biological conditions, providing durable conjugates.
In contrast to amine-reactive NHS esters (which can react with any lysine residues), maleimides offer site-selectivity when working with engineered cysteine residues or naturally occurring free thiols on biomolecules.
Read More: Water-Soluble PEG45 Linkers with NHS and Azide Groups for Therapeutic Applications
Why PEG45 as the backbone?
- Moderate hydrodynamic size: PEG45 provides ~15–16 nm of extension, reducing steric hindrance.
- Improved solubility: It increases water compatibility for hydrophobic drugs or protein complexes.
- Customizable end-groups: One end can carry a maleimide, while the other can be amine, azide, carboxyl, or methoxy for further functionalization.
PurePEG products to reference:
- mPEG45-Maleimide — methoxy-capped PEG45 with terminal maleimide.
- NH2-PEG45-Maleimide — bifunctional PEG45 with amine and maleimide termini.
- COOH-PEG45-Maleimide — carboxy-maleimide PEG45 for further conjugation flexibility.
Why Use Maleimide Linkers?
When designing PEGylated biomolecules or drug delivery systems, the maleimide functional group is often the gold standard for site-directed thiol conjugation. Here’s why:
- Exceptional Selectivity
Maleimides rarely react with other nucleophiles at physiological pH, meaning the thiol conjugation is highly predictable and clean. - Rapid Conjugation Under Mild Conditions
Reactions can occur at room temperature or slightly elevated temperatures without denaturing sensitive biomolecules. - Stable Final Product
Once formed, maleimide–thiol thioether bonds are stable, resisting hydrolysis and remaining intact through purification and storage. - Versatility for Complex Biologics
Whether you’re attaching PEG to antibodies, enzymes, or small-molecule drugs, maleimide linkers can be incorporated into modular bioconjugation workflows.
Use cases in research and therapeutics:
- Antibody-drug conjugates (ADCs): Maleimide PEG spacers enable selective drug loading at engineered cysteine sites.
- Protein labeling: PEGylation at thiols extends circulation half-life without altering protein folding.
- Enzyme modification: PEGylation shields enzymes from immune recognition while retaining catalytic activity.
Read More: Water-Soluble PEG45 Linkers with NHS and Azide Groups for Therapeutic Applications
Featured Products
Application in Thiol-Based Bioconjugation
Thiol-targeted PEGylation using maleimides is an industry workhorse because many biomolecules contain cysteine residues either naturally or engineered specifically for conjugation.
Step-by-step workflow using PEG45-Maleimide:
- Thiolation (if needed):
If your target does not naturally contain free cysteines, thiols can be introduced chemically or via mutagenesis. - Activation of the PEG linker:
PurePEG’s PEG45-Maleimide products are already pre-activated and don’t require additional reagents. - Conjugation reaction:
- Dissolve the PEG45 linker in a suitable buffer (commonly PBS or phosphate buffer, pH 6.5–7.5).
- Mix with thiolated biomolecules at stoichiometric or slight molar excess of PEG.
- Allow the reaction to proceed for 30 minutes to several hours at room temperature.
- Purification:
Use size-exclusion chromatography or ultrafiltration to remove excess PEG. - Characterization:
Confirm conjugation using MALDI-TOF, LC-MS, SDS-PAGE, or UV/Vis absorbance if PEG is tagged.
PEG45 Efficiency in Targeted Delivery Systems
Why is PEG45 often chosen over smaller PEGs (e.g., PEG4, PEG12) or larger PEGs (e.g., PEG100)?
- Intermediate size reduces immunogenicity: PEG45 is long enough to shield biomolecules from rapid clearance but not so long as to trigger immune recognition.
- Improved hydrodynamics: PEG45 increases solubility of hydrophobic drug payloads and improves overall formulation stability.
- Optimized circulation: In vivo studies show that PEGylation with intermediate-length PEG chains balances retention time and target engagement.
Case examples:
- Antibody conjugates: A PEG45 spacer prevents steric interference between antibody-binding domains and small-molecule payloads.
- Nanoparticle delivery systems: PEG45 chains create a hydration shell that prevents opsonization and aggregation.
(Product reference: Link to PurePEG’s mPEG45-Maleimide product page to highlight availability.)
Troubleshooting and Optimization
Even robust chemistries like maleimide–thiol reactions can have pitfalls. Common issues include:
- Hydrolysis of maleimide before reaction:
Maleimides can slowly hydrolyze in aqueous solution. Prepare fresh linker solutions just before use. - Over-conjugation or crosslinking:
Using too much PEG45-Maleimide may lead to unwanted protein crosslinking. Carefully control stoichiometry. - pH sensitivity:
The reaction is fastest at pH ~6.5–7.0. At higher pH, maleimide may also react with amines, reducing selectivity. - Storage considerations:
Store lyophilized PEG45-Maleimide linkers in a dry, dark environment at −20 °C for long-term stability.
Optimization tips:
- Use freshly prepared buffers.
- Remove excess reducing agents (like DTT or TCEP) before conjugation — they can quench maleimides.
- Test reaction progress with analytical HPLC or MALDI-MS to avoid overreaction.
Conclusion: Why Choose PurePEG for PEG45-Maleimide?
When high-purity PEG linkers are required, impurities or byproducts can sabotage conjugation efficiency. PurePEG provides PEG45-Maleimide linkers with exceptional quality control and lot-to-lot consistency.
- >99% purity verified by HPLC and NMR.
- Multiple formats available — methoxy, amine, and carboxyl end groups.
- Ready to ship globally, supporting research, clinical, and GMP-grade workflows.
Read More: Water-Soluble PEG45 Linkers with NHS and Azide Groups for Therapeutic Applications