Antibody-Drug Conjugates (ADCs) represent a powerful class of targeted oncology therapeutics, designed to deliver highly potent cytotoxic agents specifically to tumor cells. However, their development is often challenged by suboptimal biophysical properties, particularly poor solubility and instability. The chemical linker tethering the antibody to the payload is a critical determinant of these properties, directly influencing the ADC’s therapeutic viability.
This case study examines a common development hurdle: an ADC candidate compromised by inadequate solubility and stability. We demonstrate how the strategic implementation of a high-purity, monodisperse PEG-based linker from PurePEG’s portfolio effectively resolved these issues, transforming the conjugate into a developable drug candidate.
Introduction: The Critical Role of the Linker in ADC Design
ADC Overview and Linker Function
Antibody-drug conjugates (ADCs) constitute a sophisticated therapeutic system composed of a targeting monoclonal antibody, a cytotoxic payload, and a chemical linker. The linker serves not only as a connecting unit but also as a key functional component regulating the stability, solubility, and pharmacokinetic properties of the ADC. To ensure targeted therapeutic efficacy, ADCs must maintain structural integrity during systemic circulation for precise delivery and possess sufficient solubility to meet clinical intravenous administration requirements. Insufficient solubility readily induces molecular aggregation, potentially triggering immunogenic responses, diminished efficacy, and manufacturing challenges. Conversely, premature payload release due to linker instability can cause systemic toxicity, fundamentally undermining the strategic value of targeted therapy.
Background: The ADC Development Challenge
Project Overview and Formulation Problem
A biopharmaceutical partner encountered significant challenges with a promising ADC candidate. The molecule consisted of a validated antibody conjugated to a highly potent yet extremely hydrophobic payload. During pre-formulation studies, the ADC exhibited pronounced aggregation and precipitation, preventing its concentration to the required levels for clinical dosing.
Common Solubility and Stability Challenges
The challenges faced by the team are common in ADC development:
- Payload-Induced Aggregation: The hydrophobic nature of the payload created intermolecular interactions, leading to the formation of high molecular weight aggregates.
- Risk of Premature Payload Release: The initial linker chemistry raised concerns about stability in plasma..
- Manufacturing and Scalability Issue: Aggregation and poor solubility resulted in difficult purification, inconsistent batch quality, and challenges in process scale-up.
The initial non-PEG linker used in their construct failed to mitigate the payload’s hydrophobicity, leading to an ADC that was therapeutically promising but practically unworkable.
Featured Products
The PEG-Based Linker Solution
Rationale for PEG Linker Selection
After consulting with our experts, the team decided to re-engineer their ADC using a PEG-based heterobifunctional linker. The rationale was clear: polyethylene glycol (PEG) is a highly hydrophilic, flexible, and biocompatible polymer. By incorporating a discrete PEG chain into the linker, we can create a hydrophilic “shield” around the hydrophobic payload.
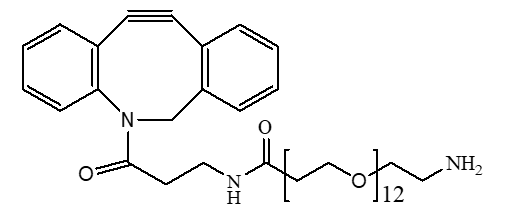
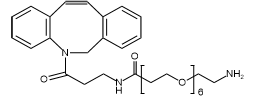
The chosen linker was a maleimide-PEG-acid, a type of Heterobifunctional PEG. This structure provided a maleimide group to specifically conjugate to a cysteine residue on the antibody and a carboxylic acid group for attaching the drug payload. The search for the “best PEG linker for protein conjugation” often leads to this kind of precise, site-specific chemistry.
Mechanism of Action: How PEG Enhances Performance
- Solubility Enhancement: The hydrophilic PEG chain significantly increases the overall aqueous solubility of the ADC.
- Stability Improvement: The flexible spacer arm reduces hydrophobic interactions between payloads on different ADC molecules, minimizing aggregation and maintaining a monodisperse formulation.
Key Features of the Selected Linker
The team selected a monodisperse Mal-PEG8-COOH from our catalog. Key features include:
- Monodispersity: With a purity of >99%, this single-molecular-weight linker ensures that every ADC molecule is identical.
- Optimal Length: The PEG8 chain provided the optimal spacing to insulate the payload without being excessively long, which could negatively impact pharmacokinetics.
- Orthogonal Reactivity: The maleimide and acid groups allow for a controlled, two-step conjugation process, ensuring a precisely constructed ADC.
Methodology and Experimental Design
Conjugation and Characterization
The conjugation was performed in a sequential manner. First, the drug payload was activated and reacted with the acid group of the Mal-PEG8-COOH linker. After purification, the maleimide-functionalized payload-linker complex was conjugated to the reduced antibody.
The final ADC was then extensively characterized using size-exclusion chromatography (SEC) to confirm aggregation levels, hydrophobic interaction chromatography (HIC) to assess hydrophobicity, and mass spectrometry to verify the drug-to-antibody ratio (DAR).
Stability and Solubility Assessments
- Solubility: The maximum concentration of the ADC that could be achieved in a standard formulation buffer without precipitation was measured.
- Stability: The ADC was incubated in human serum at 37°C for several days. Aliquots were taken at various time points and analyzed by SEC to measure the percentage of aggregated ADC and by RP-HPLC to quantify any premature drug release.
Metrics used to evaluate linker performance
The primary metrics were:
- Percentage of high molecular weight species (aggregates).
- Maximum achievable concentration (mg/mL).
- Percentage of free drug released over 7 days in serum.
Results: Significant Enhancement in ADC Properties
The data clearly demonstrated the transformative impact of the PEG-based linker.
Dramatic Solubility Improvement
The original ADC (using a non-PEG linker) could only be concentrated to 2 mg/mL before significant precipitation occurred. The new ADC, constructed with the Mal-PEG8-COOH linker, was successfully concentrated to over 20 mg/mL—a 10-fold increase, bringing it well within the range required for clinical formulation.
Superior Stability Profile
Serum stability studies revealed a substantial improvement:
- Aggregation: The PEGylated ADC showed <5% aggregation after 7 days, compared to >30% for the original construct.
- Payload Release: Premature release was reduced from 15% to <2%, confirming enhanced linker stability in circulation.
Comparative Analysis
The head-to-head comparison confirmed the hypothesis: incorporating a discrete, hydrophilic PEG linker directly addresses the solubility and stability challenges posed by hydrophobic payloads. The non-PEG linker failed to shield the payload, leading to an unviable drug candidate. The PEG linker created a stable, soluble, and highly promising therapeutic.
Discussion and Lessons Learned
Key Success Factors
This case study highlights a fundamental principle in modern drug development: the linker is not just a connector, but a critical performance-enhancing component. The PEG linker succeeded because it directly tackled the root cause of the problem—payload hydrophobicity—by introducing a dominant hydrophilic element. The success of this PEG linker for ADCs underscores the importance of rational design based on physicochemical principles.
Implications for Future ADC Development
As drug developers push the boundaries with even more complex and potent payloads, the need for advanced linkers will only grow. This study validates the strategy of using high-purity, monodisperse PEG linkers as a go-to solution for mitigating aggregation and improving stability. Approaches like a cleavable PEG linker synthesis can be combined with this principle to add payload-release functionality.
Best Practices for Linker Selection
- Thorough Payload Characterization: Understand the physicochemical properties of your drug. If it is hydrophobic, a PEG linker should be a primary consideration.
- Prioritize Monodispersity: Eliminate variability from the start by using high-purity, single-molecular-weight linkers. This simplifies analysis and de-risks your development pathway.
- Empirical Screening: The optimal PEG length can vary. It’s often wise to screen a few different lengths (e.g., PEG4, PEG8, PEG12) to find the best balance of solubility, stability, and pharmacokinetics.