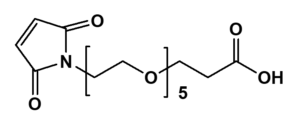
Maleimide-PEG-Acid
The Mal-PEGn-CH₂CH₂COOH series, exemplified by Mal-PEG8-CH₂CH₂COOH, is a heterobifunctional PEG derivative engineered for sequential conjugation strategies. This reagent enables thiol-specific binding through maleimide chemistry followed by carboxyl-mediated amide bond formation with amines, making it ideal for constructing complex biomolecular assemblies. Key advantages include orthogonal reactivity for versatile bioconjugation, tunable PEG spacer length (n), and minimized steric hindrance afforded by the extended ethylene chain.