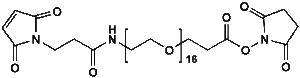
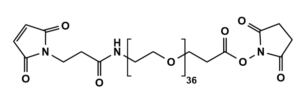
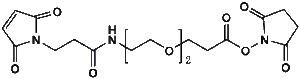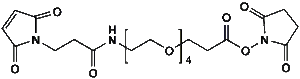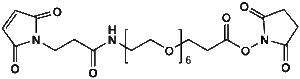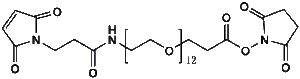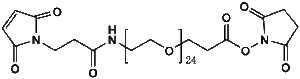Maleimide-NH-PEG-NHS ester
Mal-NH-PEGn-CH₂CH₂COONHS Ester is a heterotrifunctional reagent designed for efficient multi-step bioconjugation, enabling sequential thiol-specific maleimide coupling, amine-reactive NHS ester binding, and further functionalization via the intermediate amine group. Its key advantages include high reactivity for diverse biomolecular assembly, tunable spacer length (n), and reduced steric hindrance, though the NHS ester requires anhydrous handling due to hydrolysis sensitivity and the maleimide group may undergo retro-Michael reactions under physiological conditions.