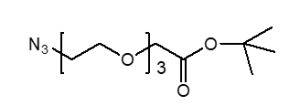
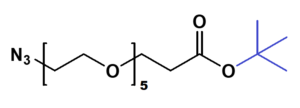
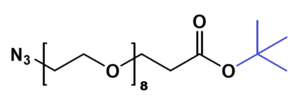
Azido-PEG-tBu
N₃-PEGn-CH₂CH₂COO-tBu is a heterobifunctional reagent designed for sequential bioorthogonal conjugation and carboxyl activation, enabling azide-alkyne click chemistry (e.g., CuAAC/SPAAC) followed by acidic deprotection of the tert-butyl ester for further carboxylic acid-mediated modifications. Its key advantages include orthogonal reactivity for controlled stepwise assembly, tunable spacer length (n), and reduced steric hindrance due to the extended chain, though the t-Bu ester requires acidic deprotection and the azide group may need protection against reduction.