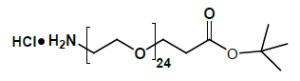
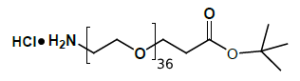
Amino-PEG-propionic acid t-butyl ester
The Amino-PEG-propionic acid t-butyl ester series, exemplified by H₂N-PEG8-CH₂CH₂COOtBu, is a heterobifunctional reagent engineered for sequential conjugation strategies. This molecule combines a primary amine group for direct amidation reactions with a tert-butyl-protected carboxylic acid group, which can be selectively activated under acidic conditions to generate the free carboxylate for subsequent functionalization. Key advantages of this series include orthogonal reactivity for controlled stepwise assembly, tunable PEG spacer length (n), and minimized steric hindrance afforded by the extended ethylene chain. It should be noted that the tert-butyl ester group requires acidic deprotection (e.g., using TFA) to liberate the carboxylic acid functionality for further conjugation.