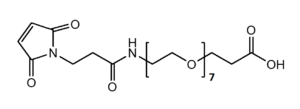
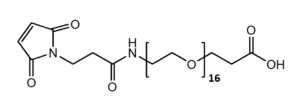
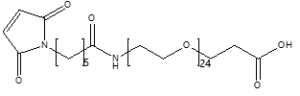
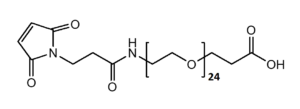
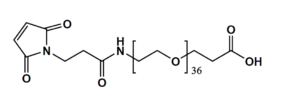
Maleimide-NH-PEG-Acid
Mal-NH-PEGn-CH₂CH₂COOH is a heterotrifunctional PEG reagent engineered for multi-step bioconjugation, enabling sequential maleimide-thiol coupling, amine-mediated amidation, and carboxyl-based modifications in oriented assemblies. Its key advantages include orthogonal reactivity for complex molecule construction, tunable spacer length (n), and enhanced solubility for hydrophobic compounds, though the maleimide group requires protection against hydrolysis and the amine may need capping during carboxyl activation to prevent cross-reactivity.