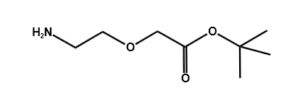
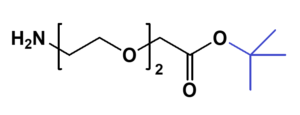
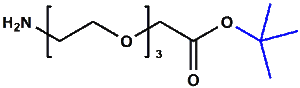
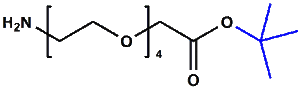
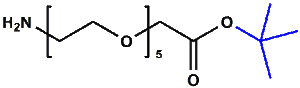
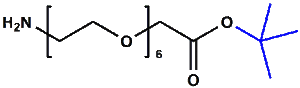
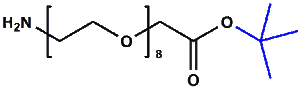
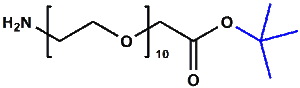
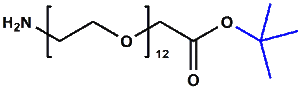
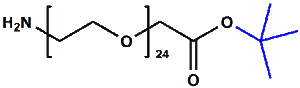
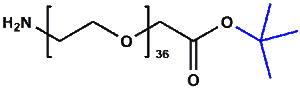
Amino-PEG-acetic acid t-butyl ester
H₂N-PEGn-CH₂COOtBu is a heterobifunctional reagent designed for sequential conjugation strategies, featuring a primary amine for immediate amidation and a protected tert-butyl ester that requires acidic deprotection to reveal a carboxylic acid for further functionalization. Its key advantages include orthogonal reactivity for controlled stepwise modifications, tunable spacer length (n), and protection of the carboxyl group during synthetic operations, though the t-Bu ester necessitates acidic conditions for deprotection.