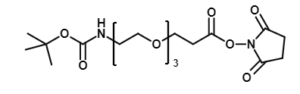
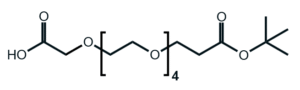
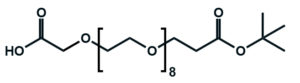
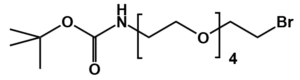
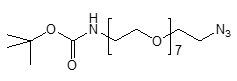
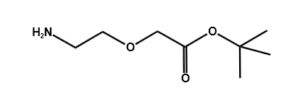
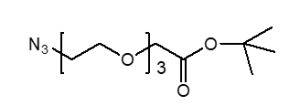
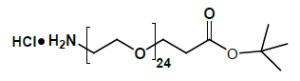
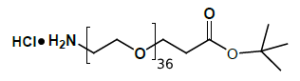
Fmoc Boc CBZ tBu
Linkers with protecting groups are bifunctional molecules featuring temporarily masked reactive sites (e.g., Fmoc, Boc, Cbz, and tBu) that can be selectively deprotected under specific conditions. They enable sequential and orthogonal conjugation in complex syntheses—such as peptide assembly, solid-phase chemistry, and prodrug development—by preventing side reactions and ensuring precise structural control. These regulated deprotection capabilities make them indispensable tools in advanced organic synthesis, bioconjugation, and therapeutic agent development.