PEG4 (PEG-4) Linkers: Structure, Protection Groups, and Applications
Polyethylene glycol (PEG) linkers are foundational tools in modern bioconjugation, offering a simple way to connect biomolecules while modifying their physicochemical properties. Among the various lengths available, the PEG4 (PEG-4) linker occupies a unique position. It provides enough spatial separation to prevent steric clashing between conjugated molecules, yet remains short enough to avoid the unpredictable folding or immunogenicity sometimes associated with much larger polymeric PEGs.
For medicinal chemists and drug delivery researchers, selecting the right spacer length is a critical optimization step. A PEG4 spacer offers a highly predictable, monodisperse structure consisting of exactly four ethylene glycol units. This exact discrete length ensures batch-to-batch consistency, which is a regulatory necessity for therapeutics like antibody-drug conjugates (ADCs) and targeted radioligands.
Understanding the specific chemistry of a PEG4 bioconjugation linker—including its available functional groups, orthogonal protection strategies, and structural advantages over other PEG lengths—is essential for designing stable, effective conjugates.
What Is a PEG4 Linker?
A PEG4 linker is a discrete, short-chain polyethylene glycol spacer containing exactly four repeating ethylene oxide units (-CH2-CH2-O-). Unlike traditional polydisperse PEG polymers, which consist of a mixture of different chain lengths and molecular weights, discrete PEG4 linkers have a single, defined molecular weight.
PEG4 structure and spacer length
A PEG4 spacer provides an approximate maximum extended length of ~14 Å, although its effective distance in solution may vary depending on molecular conformation and terminal functional groups. This specific spacer length physically separates the two conjugated entities, such as a fluorophore and a protein, or a cytotoxic payload and a monoclonal antibody. The oxygen atoms within the ethylene glycol backbone readily form hydrogen bonds with water molecules, imparting high hydrophilicity to the entire construct.
Why PEG4 is commonly used in bioconjugation
Chemists frequently choose the PEG4 PEG linker because it perfectly balances solubility enhancement with structural compactness. Many small-molecule drugs and fluorescent dyes are highly hydrophobic. When conjugated directly to a protein, these hydrophobic molecules can force the protein to misfold or aggregate. Inserting a PEG4 spacer masks this hydrophobicity, keeping the final bioconjugate soluble in aqueous buffers without adding excessive bulk to the molecule.
PEG4 vs longer PEG spacers
While longer PEG spacers (like PEG12 or PEG24) offer massive increases in water solubility, they can also introduce complications. Very long PEG chains have a high hydrodynamic radius and can wrap around the target protein, potentially blocking active sites or binding interfaces. Longer PEGs can also trigger anti-PEG antibody responses in vivo. A PEG-4 linker avoids these issues, providing just enough hydrophilicity and spatial separation without wrapping or shielding critical protein domains.
PEG4 Linker Structure and Functional Groups
To facilitate specific bioconjugation reactions, the ends of the PEG4 chain are functionalized with reactive moieties. These groups dictate which amino acid residues or chemical tags the linker will react with.
Amine PEG4 linkers
Amine-terminated PEG4 linkers contain a primary amine (-NH2) at one or both ends. These are typically used in reactions with activated esters (such as NHS esters) or carboxylic acids in the presence of coupling reagents like EDC and NHS. Amine PEG4 linkers are highly stable and are frequently used to modify surfaces or attach PEG chains to carboxyl-rich biomolecules.
Carboxyl PEG4 linkers
Carboxyl PEG4 linkers feature a terminal carboxylic acid (-COOH). To react with target amines, the carboxyl group must first be activated. Chemists typically convert the carboxyl group into a reactive NHS ester or use carbodiimide chemistry (EDC/sulfo-NHS) directly in the reaction buffer. This approach is standard for labeling peptides and proteins at their N-terminus or surface lysine residues.
NHS PEG4 linkers
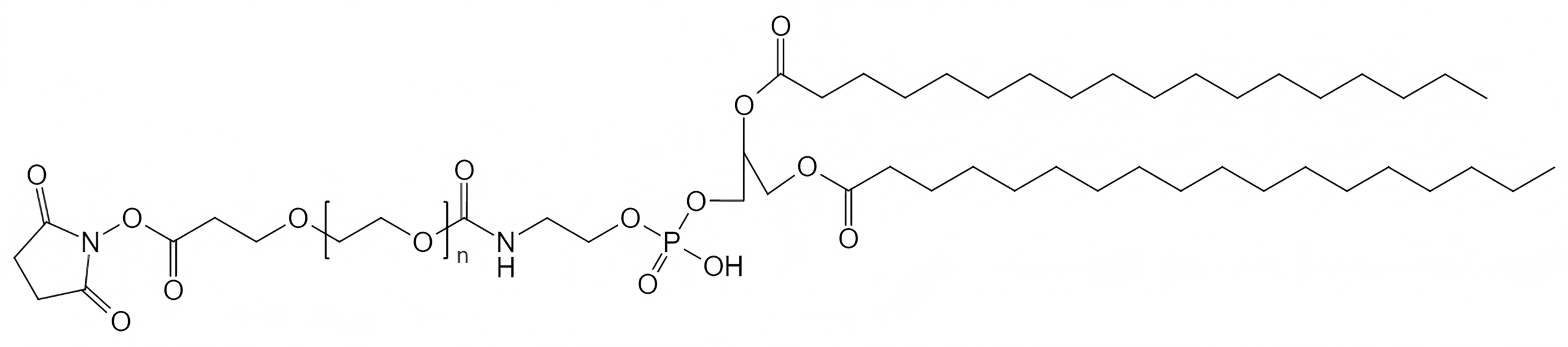
An NHS PEG4 linker is pre-activated and ready to react with primary amines at physiological pH (7.2 to 8.0). The N-hydroxysuccinimide (NHS) ester undergoes nucleophilic acyl substitution when exposed to amines, forming a stable amide bond. NHS-PEG4 linkers are highly favored for antibody labeling because antibodies possess numerous accessible lysine residues.
Maleimide PEG4 linkers
When site-specific conjugation is required, maleimide PEG4 linkers are the standard choice. The maleimide group reacts highly selectively with free sulfhydryls (thiols) at a slightly acidic to neutral pH (6.5 to 7.5), forming a stable thioether bond. This chemistry is heavily utilized in attaching payloads to the hinge-region cysteines of monoclonal antibodies.
Azide and alkyne PEG4 linkers
For bioorthogonal click chemistry, azide and alkyne PEG4 linkers are indispensable. A PEG4 azide will react with a terminal alkyne via Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC), or with a strained alkyne (like DBCO) via Strain-Promoted Azide-Alkyne Cycloaddition (SPAAC). These reactions occur rapidly and specifically in complex biological environments without interfering with native biological functionalities.


Protection Groups Used in PEG4 Linkers
In complex multi-step organic synthesis, reactive functional groups on a heterobifunctional PEG4 linker must be temporarily masked to prevent unwanted side reactions. This is achieved using temporary chemical covers known as protection groups.
Boc-protected PEG4 linkers
The tert-butyloxycarbonyl (Boc) group is a widely used protecting group for primary amines. A Boc PEG4 linker keeps the amine masked while reactions are performed on the opposite end of the PEG chain. The Boc group is highly stable under basic and nucleophilic conditions. Cleavage of the Boc group requires strong acidic conditions, most commonly pure trifluoroacetic acid (TFA) or a mixture of TFA and dichloromethane (DCM).
Fmoc-protected PEG4 linkers
The fluorenylmethyloxycarbonyl (Fmoc) group is another standard amine-protecting group, particularly dominant in solid-phase peptide synthesis (SPPS). An Fmoc PEG4 linker is stable under acidic conditions but is rapidly cleaved by mild organic bases. Standard deprotection protocols utilize 20% piperidine in dimethylformamide (DMF), which removes the Fmoc group and exposes the free amine for the next coupling step.
Boc vs Fmoc for PEG4 chemistry
Choosing between a Boc vs Fmoc PEG4 linker depends entirely on the orthogonal stability required for the synthesis pathway. If the molecule contains acid-sensitive moieties (such as certain glycosidic bonds or acetals), a Boc-protected PEG4 linker should be avoided, and an Fmoc-protected linker should be used instead. Conversely, if the synthesis involves strong base-catalyzed steps, Boc is the necessary choice to ensure the amine remains safely protected.
When protection groups are required
Protection groups are mandatory when synthesizing heterobifunctional PEG4 linkers from homobifunctional precursors, or when building complex targeting ligands step-by-step. Without a protected PEG4 linker, the molecule could polymerize with itself, or the reactive groups could be destroyed by the reagents used in intermediate synthetic steps.
Why PEG4 Is Used as a Spacer in Bioconjugation
The physical and chemical properties of the PEG4 spacer make it a highly effective tool for solving common bioconjugation challenges.
Improving solubility
Many fluorescent dyes (like cyanines) and cytotoxic payloads (like auristatins) are highly lipophilic. When conjugated directly to a biomolecule, they drastically reduce the aqueous solubility of the construct. The strong hydrogen-bonding capacity of the PEG4 bioconjugation linker counteracts this lipophilicity, keeping the conjugate fully dissolved in physiological buffers and preventing premature clearance in vivo.
Reducing steric hindrance
Steric hindrance occurs when two bulky molecules are forced too close together, impeding their ability to interact with their respective targets. If a drug is attached flush against a carrier protein, the protein’s bulky tertiary structure may block the drug from entering a target receptor. The PEG4 spacer pushes the conjugated molecule away from the protein surface, giving it the rotational freedom required to bind its target.
Controlling linker distance
Certain biological interactions require precise spatial geometries. In bispecific engagers or PROTACs (Proteolysis Targeting Chimeras), the distance between the two binding domains dictates the efficacy of the induced protein-protein interaction. A PEG4 spacer provides a rigid, predictable distance that can be systematically lengthened or shortened (to PEG3 or PEG5) during structure-activity relationship (SAR) studies.
Improving conjugation efficiency
By moving the reactive functional group away from the bulky core of the payload or the protein surface, a PEG4 spacer reduces local steric crowding at the reaction site. This makes the reactive moiety (such as an NHS ester or maleimide) much more accessible to the incoming nucleophile, significantly increasing overall conjugation yields and reducing reaction times.
PEG4 vs PEG2 vs PEG8: Spacer Length Comparison
Selecting the ideal discrete PEG length requires balancing solubility, distance, and molecular weight.
When PEG4 is preferred
PEG4 is the preferred default spacer for general bioconjugation. It offers enough distance to relieve steric hindrance and enough oxygen atoms to provide a noticeable solubility boost, without drastically altering the molecular weight or pharmacokinetic profile of the carrier protein.
When shorter PEG spacers work better
A PEG2 linker is chosen when minimal distance is required, or when the overall molecular weight of the conjugate must be kept as low as possible. PEG2 provides very little solubilization power compared to PEG4. It is strictly used as a minimal spacer to separate a reactive group from a payload just enough to allow conjugation chemistry to proceed.
When longer PEG spacers are required
A PEG8 linker (or longer) is utilized when conjugating exceptionally hydrophobic molecules, such as certain pyrrolobenzodiazepine (PBD) dimers used in ADCs. The extra ethylene glycol units are necessary to prevent the hydrophobic payloads from driving protein aggregation. PEG8 is also used when a very long physical reach is required to penetrate deep binding pockets on cell surface receptors.


PEG4 Linkers in Antibody Drug Conjugates
The development of modern ADCs relies heavily on optimized linker chemistry to deliver potent cytotoxins to tumor cells safely.
Why PEG4 is used in ADC design
In ADC design, the linker must keep the cytotoxic payload securely attached while in systemic circulation, yet release it efficiently once internalized by the target cell. A PEG4 linker ADC strategy is often employed to attach the payload to a cleavable peptide sequence (like Val-Cit). The PEG4 spacer ensures the peptide sequence is exposed to cleavage enzymes (like Cathepsin B) in the lysosome, rather than buried against the antibody surface.
PEG4 and payload spacing
Cytotoxic payloads like MMAE are bulky. If conjugated too close to the antibody, the steric bulk can physically disrupt the antibody’s paratope, reducing its binding affinity to the tumor antigen. The PEG4 spacer acts as a flexible tether, distancing the drug from the antibody structure and preserving full antigen-binding capacity.
PEG4 impact on solubility
High Drug-to-Antibody Ratios (DAR) are desirable for ADC potency but are notoriously difficult to achieve with hydrophobic payloads. Piling multiple hydrophobic molecules onto an antibody causes rapid aggregation and clearance by the liver. Integrating a PEG-4 linker adjacent to the payload masks its hydrophobicity, allowing chemists to achieve higher DARs (e.g., DAR 4 or DAR 8) without compromising the structural integrity or solubility of the ADC.
Common Types of PEG4 Linkers
Depending on the intended reaction, chemists utilize different structural classes of PEG4 linkers.
Heterobifunctional PEG4 linkers
Heterobifunctional PEG4 linkers contain two entirely different reactive groups at opposite ends of the chain (e.g., Azide-PEG4-Amine or NHS-PEG4-Maleimide). These are the most valuable linkers in bioconjugation, as they allow for highly controlled, directional, two-step conjugation procedures without the risk of cross-linking.
Homobifunctional PEG4 linkers
Homobifunctional PEG4 linkers have the same reactive group on both ends (e.g., NHS-PEG4-NHS). These are typically used as crosslinkers to covalently bind two of the same proteins together, or to stabilize multi-subunit protein complexes for structural analysis.
Cleavable PEG4 linkers
Cleavable PEG4 linkers incorporate a specific trigger—such as a disulfide bond (cleavable by reduction), a hydrazone (cleavable by acid), or a peptide sequence (cleavable by proteases). These are critical in drug delivery to ensure the active payload is released precisely upon entering the target microenvironment.
Non-cleavable PEG4 linkers
Non-cleavable PEG4 linkers rely on stable amide or thioether bonds that resist degradation in the bloodstream and the lysosome. When an ADC utilizes a non-cleavable PEG4 linker, the payload is only released after the entire antibody is completely catabolized into individual amino acids within the target cell.
Related Products
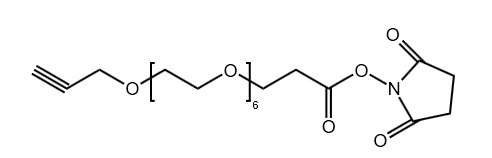
Propargyl-PEG6-NHS Ester
CAS# 2093153-99-0
MW# 489.52
5,15-bis(4-aminophenyl)-10,20-diphenylporphyrin-MnCl
CAS# N/A
MW# 733.15
5-(4-carboxyphenyl)-10,15,20-triphenyl porphyrin-MnCl
CAS# N/A
MW# 747.13
5-(4-isothiocyanatophenyl)-10,15,20-(triphenyl)porphyrin
CAS# 863708-54-7
MW# 671.82
5,15-Di(4-aminophenyl)-10,20-diphenyl porphine
CAS# 116206-75-8
MW# 644.78
How to Choose the Right PEG4 Linker
Selecting the exact PEG4 linker requires an assessment of the target molecules and the desired reaction environment.
Functional group compatibility
First, audit the available chemical handles on your target biomolecules. If you are targeting lysines, choose an NHS PEG4 linker. If you are targeting a reduced hinge cysteine on an antibody, select a maleimide PEG4 linker. Ensure the two groups on a heterobifunctional linker do not cross-react.
Spacer length considerations
Verify that a ~15 angstrom distance is appropriate for your application. If you require a tighter fit for a PROTAC, you may need to drop to a PEG3. If your fluorophore is quenching due to proximity to a metal center, a PEG4 spacer is usually sufficient to restore fluorescence.
Solubility requirements
Assess the hydrophobicity of your payload. If your payload is moderately lipophilic, PEG4 will likely resolve solubility issues in an aqueous buffer. If you are observing immediate precipitation upon conjugation, you may need to switch to a PEG8 or PEG12 linker to force the payload into solution.
Stability considerations
Consider the pH of your conjugation buffer. NHS esters degrade rapidly at high pH, requiring immediate use. Maleimides can undergo ring-opening hydrolysis over time. Plan your synthetic route to accommodate the stability half-life of the reactive groups on your chosen PEG-4 linker.

Selecting the Optimal Reagents for Bioconjugation
Choosing the exact PEG linker dictates the physical stability, solubility, and ultimately the biological success of a conjugate. By understanding the distinct advantages of the PEG4 structure, orthogonal protection strategies, and appropriate reactive groups, researchers can design more stable and highly targeted therapeutic constructs.
Frequently Asked Questions
PEG4 refers to a discrete polyethylene glycol chain composed of exactly four repeating ethylene oxide (-CH2-CH2-O-) units. This provides a specific, uniform molecular weight rather than a mixture of polymer lengths.
A PEG4 linker is used primarily as a molecular spacer in bioconjugation to link two molecules together (like a drug and an antibody). It provides an optimal distance to prevent steric hindrance while improving the water solubility of hydrophobic payloads.
PEG4 is used instead of PEG8 when a shorter spatial distance is required, or when the extreme solubility profile of PEG8 is unnecessary. PEG4 keeps the overall molecular weight lower and avoids potential issues with long PEG chains wrapping around active sites.
Boc-PEG4 is a PEG4 linker where one of the reactive amine groups is temporarily masked by a tert-butyloxycarbonyl (Boc) protecting group. It is highly stable in basic conditions and is removed using strong acids like TFA.
Fmoc-PEG4 is a PEG4 linker protected by a fluorenylmethyloxycarbonyl (Fmoc) group. It is stable in acidic environments but is easily cleaved by mild organic bases, making it standard for solid-phase peptide synthesis.