Introduction to PEG Linkers in Drug Design
In the complex world of drug development, a linker is the unsung hero that connects a therapeutic payload to the payload’s delivery vehicle. The properties of this molecular bridge can make or break a drug’s success. Choosing the right linker is critical, and a key consideration is its relationship with water—whether it is hydrophilic or hydrophobic.
This guide explores the distinct roles of hydrophilic and hydrophobic PEG linkers in drug design. We will examine their chemical properties, unique advantages, and how their differences impact a drug’s behavior in the body.
What Are PEG Linkers?
PEG linkers are synthetic polymers composed of repeating ethylene glycol units that serve as stable yet flexible molecular spacers in bioconjugation. The process of covalently attaching these PEG chains to therapeutic molecules, known as PEGylation, is a well-established strategy for enhancing key drug properties. These linkers primarily function by:
- providing optimal spatial separation to improve binding orientation and accessibility;
- enhancing physicochemical characteristics including solubility and stability while reducing aggregation;
- modulating pharmacokinetic profiles by prolonging circulation half-life and altering biodistribution patterns.
At PurePEG, we specialize in high-purity, monodisperse PEGs, providing the consistency required for robust and reproducible drug development.
Hydrophilic PEG Linkers
Characteristics: The ether oxygen atoms in the PEG backbone form hydrogen bonds with water, granting these linkers high aqueous solubility.
Key Advantages & Applications:
- Enhanced Solubility:They dramatically improve the water solubility of hydrophobic drug payloads, enabling viable intravenous formulations. Our PEGylation Reagents are specifically designed for this purpose.
- Improved Stability & Extended Half-life: The flexible PEG chain creates a protective hydration shell, shielding the drug from enzymatic degradation and immune recognition. The increased hydrodynamic size also reduces renal clearance, leading to a longer circulation half-life. Products like our PEG45 series are ideal for achieving sustained systemic exposure.
- Ideal for Biologics & ADCs: In complexes like ADCs, a hydrophilic PEG linker ensures the conjugate remains soluble and stable in the bloodstream. Our range of Heterobifunctional PEGs provides the necessary functionality and hydrophilicity to optimize pharmacokinetic profiles.
When to Use: Choose hydrophilic PEG linkers when your primary goals are improving aqueous solubility, increasing systemic circulation time, and enhancing stability for biologics and targeted delivery systems.
Hydrophobic PEG Linkers
Characteristics: These linkers incorporate non-polar segments (e.g., alkyl chains) that favor lipid environments over aqueous ones.
Key Advantages & Applications:
- Facilitated Cellular Uptake:Their lipid-compatible nature aids in crossing the cell membrane, which is crucial for drugs with intracellular targets.
- Core Component of Nanocarriers: In lipid nanoparticles (LNPs) and polymeric micelles, hydrophobic segments are essential for encapsulating poorly soluble drugs. Our PEG-Lipid products combine a hydrophilic PEG corona with a hydrophobic lipid anchor, forming the foundational building blocks of stable, long-circulating nanocarriers.
- Critical for PROTAC Design:In Proteolysis-Targeting Chimeras (PROTACs), the linker must facilitate the formation of a productive ternary complex. The rigidity and moderate hydrophobicity of the linker can be crucial for proper orientation and efficacy. Explore our specialized PROTAC Linkers for these advanced applications.
When to Use: Opt for hydrophobic linkers or hybrid PEG-lipids when your application requires cell membrane penetration, intracellular targeting, or the assembly of nanoparticle-based delivery systems
Key Differences Between Hydrophilic and Hydrophobic PEG Linkers
The choice between a hydrophilic and a hydrophobic linker fundamentally alters a drug’s in vivo behavior.
Caption: Visual showing how hydrophilic linkers improve solubility in water, while hydrophobic linkers interact with lipid membranes.
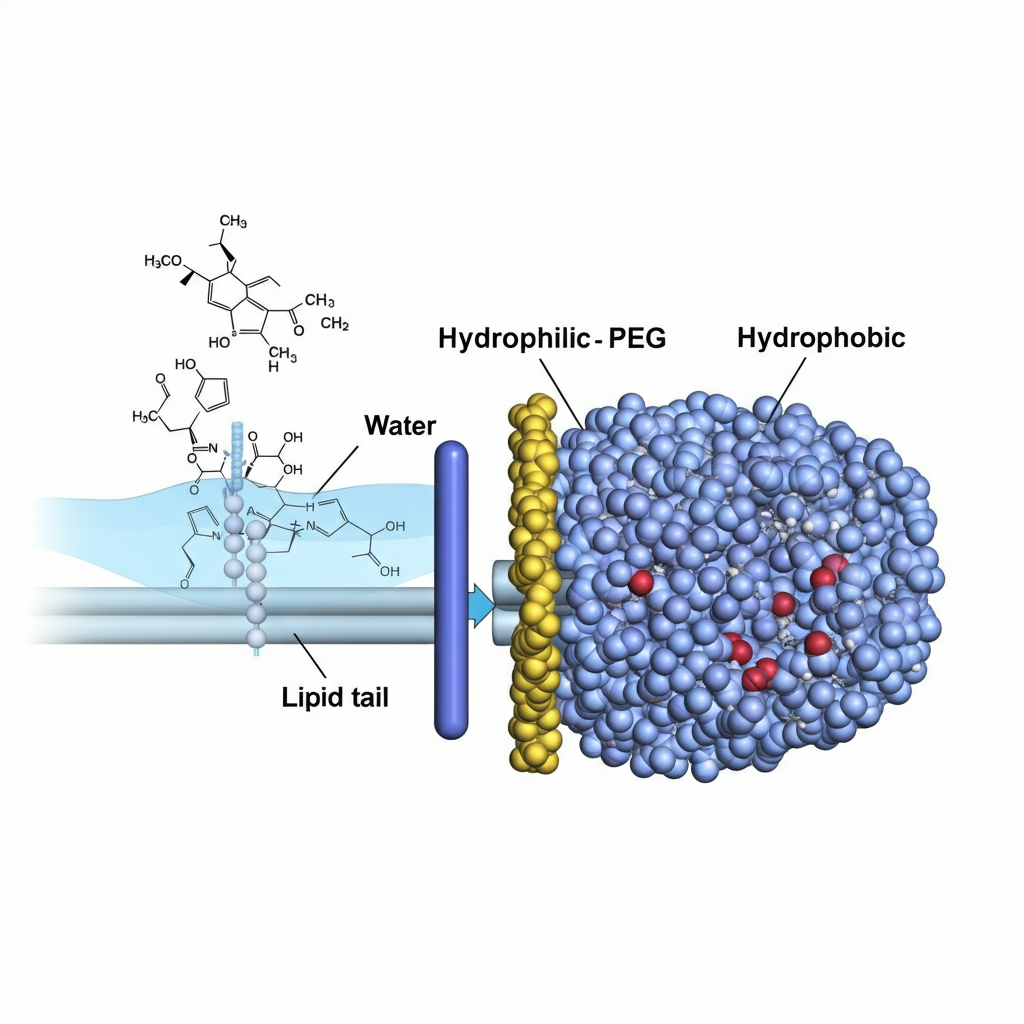
Solubility and compatibility with drugs
A hydrophilic linker is ideal for aqueous formulations and improving the solubility of hydrophobic payloads. In contrast, a hydrophobic linker might be chosen when the payload is already soluble or when the final product is a non-aqueous formulation. Combining the two, as seen in DSPE-PEG products, enables the formation of structures that can carry hydrophobic drugs within a soluble nanoparticle.
Impact on pharmacokinetics and bioavailability
Hydrophilic PEGs generally extend a drug’s circulation half-life by reducing renal clearance. Hydrophobic linkers, on the other hand, can sometimes potentially lead to faster clearance by the liver or aggregation in the bloodstream if not formulated correctly. However, their ability to facilitate effective membrane crossing can boost the bioavailability of drugs designed to target intracellular components.
Safety and immunogenicity considerations
PEG is widely recognized for its non-immunogenic properties. Its hydrophilic nature helps “hide” the attached drug from the immune system. Purely hydrophobic linkers can sometimes be perceived as foreign substances by the body and may trigger an immune response or non-specific binding to plasma proteins. Therefore, a careful balance is often required.
Conclusion: Hydrophilic vs Hydrophobic PEG Linkers
Hydrophilic PEG linkers are the cornerstone for enhancing solubility, stability, and pharmacokinetics in systemic delivery.
Hydrophobic elements are specialized tools for enabling intracellular delivery and constructing advanced nanocarriers.
The “best” linker is the one best suited to your specific application. Start by defining your core therapeutic goal and the properties of your molecule. By understanding the fundamental differences between these two linker types, you can make an strategically informed decision to drive your research forward.

