PEGylation is one of the most widely used strategies for modifying biomolecules in modern drug development. Using specialized PEGylation reagents, researchers can control how proteins, peptides, and nanoparticles behave in biological systems. From improving circulation time to enhancing stability, PEGylation plays a central role in bioconjugation and drug delivery design.
What Is PEGylation?
PEGylation refers to the covalent attachment of polyethylene glycol (PEG) chains to biomolecules such as proteins, peptides, antibodies, or small molecules.
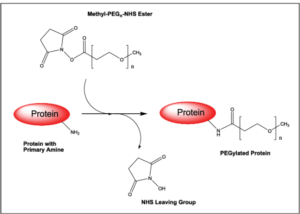
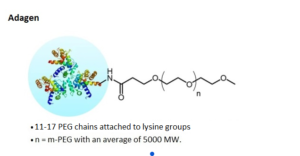
This is typically achieved by reacting an activated PEG reagent with a functional group on the target molecule under controlled conditions.
The result is a conjugate where the PEG chain becomes an integral part of the molecule’s structure, not just a coating or excipient.
Why PEGylation Is Used
PEGylation is widely applied because it consistently improves key properties of biomolecules that would otherwise limit their therapeutic use.
Improved Pharmacokinetics
PEG increases the effective size of a molecule, reducing renal clearance and extending circulation time.
Enhanced Stability
PEG chains provide steric protection against enzymatic degradation and aggregation, helping maintain structural integrity in biological environments.
Increased Solubility
Hydrophobic or poorly soluble molecules often become more water-compatible after PEGylation, simplifying formulation and delivery.
Reduced Immunogenicity
The PEG layer can shield epitopes and reduce immune recognition, although this effect depends on PEG density and exposure.
What PEGylation Changes at the Molecular Level
PEGylation is not just a surface modification. It alters multiple physicochemical properties simultaneously:
- Increased hydrodynamic radius
- Changes in molecular conformation
- Increased hydrophilicity
- Steric shielding of reactive or binding sites
These changes directly influence how the molecule interacts with proteins, cells, and clearance pathways.
PEGylation Chemistry and Reagents
PEGylation relies on selective reactions between functional groups on PEG and the target molecule.
PEG reagents can be:
- Homo-functional (same group on both ends)
- Hetero-functional (different reactive groups for controlled conjugation)
The choice of chemistry determines selectivity, stability, and reaction conditions.
PEGylation Chemistry Overview
The most commonly used PEGylation chemistries differ in selectivity, reaction conditions, and practical applications.
| Chemistry | Target Group | Selectivity | Typical Conditions | Notes |
| NHS ester | Amine | Low–medium | pH 7–9 | Fast, common |
| Maleimide | Thiol | High | pH 6.5–7.5 | Site-specific |
| Click chemistry | Azide/alkyne | Very high | Mild, aqueous-compatible | Bioorthogonal |
NHS Ester Reactions (Amine Targeting)
NHS-activated PEG reacts efficiently with primary amines on proteins (lysine residues or N-terminus).
- Optimal pH: ~7–9
- Forms stable amide bonds
- Fast and widely used
- Limited site specificity
Maleimide Reactions (Thiol Targeting)
Maleimide-PEG reacts selectively with sulfhydryl groups (cysteine residues).
- Optimal pH: ~6.5–7.5
- Forms stable thioether linkages
- Higher selectivity than NHS chemistry
- Requires accessible thiol groups
Click Chemistry (Bioorthogonal Reactions)
Click reactions such as azide-alkyne or tetrazine-TCO provide highly selective conjugation.
- No or minimal side reactions
- Can proceed in aqueous systems
- Useful for complex or sensitive biomolecules
These reactions are increasingly used when precise control over conjugation is required.
PEGylation in Therapeutic Applications
PEGylation has been applied across multiple therapeutic modalities, often solving fundamental limitations in drug design.
Protein and Antibody Therapeutics
PEGylation has been used to extend the half-life of protein drugs by reducing clearance and protecting against degradation.
This allows:
- Less frequent dosing
- Improved stability in circulation
- Enhanced therapeutic exposure
However, excessive PEGylation can reduce biological activity by blocking binding sites, so optimization is critical.
Peptide-Based Drugs
Peptides often suffer from rapid degradation and short half-life.
Attaching PEG chains can:
- Increase stability against enzymatic breakdown
- Reduce renal filtration
- Improve overall bioavailability
Even relatively small PEG chains can produce meaningful improvements in performance.
Nanoparticles and Drug Delivery Systems
PEGylation is a core strategy in nanoparticle design, particularly for lipid-based systems.
PEG on nanoparticle surfaces:
- Improves stability during formulation and storage
- Reduces aggregation
- Controls interactions with biological systems
In lipid nanoparticles, PEGylation also enables controlled surface behavior, including eventual dissociation to allow cellular uptake.
Selecting the Right PEGylation Strategy
Effective PEGylation depends on multiple variables:
- Target molecule structure
- Available functional groups
- Desired site specificity
- PEG molecular weight and architecture
- Intended biological application
There is no single “best” PEGylation approach.
The optimal design balances stability, activity, and reproducibility.
In applications where precision matters, access to well-defined, structurally consistent PEG reagents can significantly improve reproducibility and reduce variability across batches.
The Importance of PEG Structure and Purity
As PEGylation becomes more precise, the quality of PEG reagents becomes increasingly important.
Variations in PEG structure, molecular weight distribution, or functionalization can lead to:
- Inconsistent conjugation
- Batch-to-batch variability
- Changes in biological performance
Using well-defined PEG reagents helps reduce these variables and supports more reproducible outcomes, particularly in complex systems such as biologics and nanoparticle formulations.
Frequently Asked Questions
What is PEGylation used for?
PEGylation is used to improve the stability, solubility, and circulation time of biomolecules, particularly in therapeutic and drug delivery applications.
What functional groups are commonly used in PEGylation?
Common functional groups include NHS esters (amine targeting), maleimides (thiol targeting), and click chemistry groups such as azides and alkynes.
Does PEGylation affect biological activity?
It can. While PEGylation improves stability and circulation, it may reduce activity if it interferes with binding sites. Careful design is required to balance these effects.
What is the difference between site-specific and random PEGylation?
Site-specific PEGylation targets defined locations on a molecule, leading to more uniform products. Random PEGylation modifies multiple sites and can result in heterogeneous mixtures.
Final Thoughts
PEGylation remains a foundational tool in modern bioconjugation and drug delivery. Its value lies not just in extending circulation time, but in enabling precise control over how molecules behave in complex biological environments.
As systems become more advanced, the focus is shifting from simply adding PEG to designing PEGylation with intention, where structure, chemistry, and application are tightly aligned.
References
- Hou, X., et al. Lipid nanoparticles for mRNA delivery. Nature Reviews Materials (2021)
- Veronese, F.M., Pasut, G. PEGylation: successful approach to drug delivery. Drug Discovery Today (2005)
- Kanwaldeep, G., et al. PEG-lipid micelles as drug carriers. Journal of Drug Targeting (2015)

