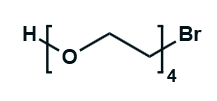
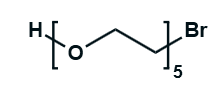
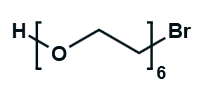
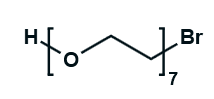
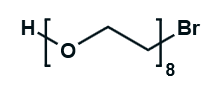
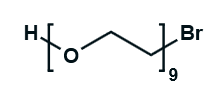
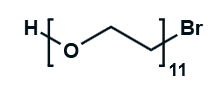
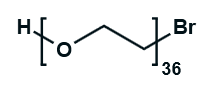
Hydroxy-PEG-Bromide
HO-PEGn-Br is a heterobifunctional PEG derivative featuring a hydroxyl group and a bromine terminus, enabling sequential conjugation strategies such as nucleophilic substitution with thiols or amines followed by hydroxyl-directed modifications. Its key advantages include orthogonal reactivity for flexible biomolecular assembly, tunable spacer length (n), and cost-effective synthesis, though the bromide group is moisture-sensitive and may require anhydrous handling to prevent hydrolysis.