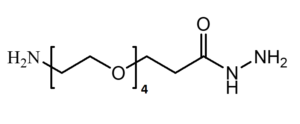
Amino-PEG-Hydrazide
H₂N-PEGn-Hydrazide is a heterobifunctional reagent designed for dual conjugation strategies, enabling sequential modifications such as amide bond formation with carboxylates followed by hydrazone linkage with carbonyl groups (e.g., in glycoproteins or oxidized antibodies). Its key advantages include orthogonal reactivity for versatile biomolecular assembly, tunable spacer length (n), and enhanced solubility, though the hydrazide group may require protection during amine-directed reactions to avoid cross-reactivity.